10.13.14
The results are in: Two sets of data from studies evaluating Sorin Group’s Perceval valve have come back with satisfying numbers.
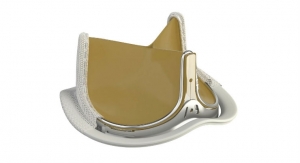
First, the the Cavalier multi-center European trial: It was designed to evaluate the safety and effectiveness of the Perceval valve in a large population requiring aortic valve replacement (AVR) with or without concomitant coronary bypass grafting. There were 658 patients included from 25 selected European centers. The 30-day results, which were presented at the European Association for Cardio-Thoracic Surgery in Milan, Italy on Oct. 13, demonstrated the safety of the valve via various criteria including reproducibility of procedure.
A second study, also presented at the conference, detailed clinical and hemodynamic outcomes up to five years in more than 700 patients. According to the presentation, Perceval demonstrated stable results at up to five years follow-up. No valve migrations, structural valve degeneration or valve thromboses were observed in the largest patient cohorts ever implanted with sutureless valves. Low early-30-day mortality (1.9 percent), low major early paravalvular leak (1.4 percent) and low early stroke (1.6 percent) rates confirmed the safety of the sutureless valve. Another noteworthy clinical benefit was the extremely low endocarditis rate (0.3 percent). This is particularly important for patient outcomes as infective endocarditis is a serious and frequently fatal condition, particularly when it occurs on a valve replacement device.
“Even in this elderly patient cohort with 40 percent of patients aged over 80 years old, early and late mortality were very low,” said Professor Malakh Shrestha, MHH, who presented the second study. “The sutureless technique is a promising alternative to stented biological aortic valve replacement.”
Based in Milan, Italy, Sorin Group makes devices for cardiovascular disease.
First, the the Cavalier multi-center European trial: It was designed to evaluate the safety and effectiveness of the Perceval valve in a large population requiring aortic valve replacement (AVR) with or without concomitant coronary bypass grafting. There were 658 patients included from 25 selected European centers. The 30-day results, which were presented at the European Association for Cardio-Thoracic Surgery in Milan, Italy on Oct. 13, demonstrated the safety of the valve via various criteria including reproducibility of procedure.
A second study, also presented at the conference, detailed clinical and hemodynamic outcomes up to five years in more than 700 patients. According to the presentation, Perceval demonstrated stable results at up to five years follow-up. No valve migrations, structural valve degeneration or valve thromboses were observed in the largest patient cohorts ever implanted with sutureless valves. Low early-30-day mortality (1.9 percent), low major early paravalvular leak (1.4 percent) and low early stroke (1.6 percent) rates confirmed the safety of the sutureless valve. Another noteworthy clinical benefit was the extremely low endocarditis rate (0.3 percent). This is particularly important for patient outcomes as infective endocarditis is a serious and frequently fatal condition, particularly when it occurs on a valve replacement device.
“Even in this elderly patient cohort with 40 percent of patients aged over 80 years old, early and late mortality were very low,” said Professor Malakh Shrestha, MHH, who presented the second study. “The sutureless technique is a promising alternative to stented biological aortic valve replacement.”
Based in Milan, Italy, Sorin Group makes devices for cardiovascular disease.