Sense Biodetection Ltd.06.15.20
Sense Biodetection Limited (Sense) has appointed John Bishop as non-executive director. Bishop was CEO of Cepheid from 2002 until its acquisition by Danaher for $4 billion in 2016. In this role he was instrumental in Cepheid’s transformation and rapid growth into a class leading molecular diagnostics company and oversaw the successful launch and commercialization of its GeneXpert and GeneXpert Xpress systems.
As a diagnostics industry leader with more than 50 years of experience in the field, Bishop will perform a pivotal strategic role at Sense as the company rapidly scales up production and distribution of its first products, including its COVID-19 test, Veros SARS-CoV-2.
Commenting on his appointment, Bishop said: "The COVID-19 pandemic has highlighted the urgent global need for broadly disseminated point of care molecular diagnostic tests, such as Sense’s instrument-free Veros, to help enable infectious disease outbreaks to be contained. I look forward to supporting the company through the scale-up and commercialisation of its first products."
Sense CEO Harry Lamble commented: "I am thrilled to welcome John Bishop to Sense’s board. His remarkable expertise and experience will greatly support our efforts to launch the world’s first instrument-free molecular diagnostic test for COVID-19, and to shape our future product pipeline."
Earlier this spring, Sense Biodetection announced an accelerated program to launch the world’s first instrument-free, point-of-care molecular diagnostic test for SARS-CoV-2, the coronavirus responsible for the COVID-19 pandemic.
Sense is partnering closely with Phillips-Medisize, a Molex company and leading global medical device innovator, developer and manufacturer, to scale-up production of its test in order to meet the growing demand for rapid diagnostics.
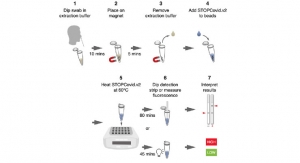
Sense’s Veros SARS-CoV-2 is a simple disposable test that uses a nasal swab sample to give an ultra-rapid result without the need for any instrumentation. As a molecular test its performance is equivalent to Gold Standard laboratory tests but it is easy to use in any setting and results are available in under 10 minutes. The test is fully self-contained and can be widely distributed to wherever it is needed, overcoming the logistical and contamination problems associated with machine-based testing.
As a diagnostics industry leader with more than 50 years of experience in the field, Bishop will perform a pivotal strategic role at Sense as the company rapidly scales up production and distribution of its first products, including its COVID-19 test, Veros SARS-CoV-2.
Commenting on his appointment, Bishop said: "The COVID-19 pandemic has highlighted the urgent global need for broadly disseminated point of care molecular diagnostic tests, such as Sense’s instrument-free Veros, to help enable infectious disease outbreaks to be contained. I look forward to supporting the company through the scale-up and commercialisation of its first products."
Sense CEO Harry Lamble commented: "I am thrilled to welcome John Bishop to Sense’s board. His remarkable expertise and experience will greatly support our efforts to launch the world’s first instrument-free molecular diagnostic test for COVID-19, and to shape our future product pipeline."
Earlier this spring, Sense Biodetection announced an accelerated program to launch the world’s first instrument-free, point-of-care molecular diagnostic test for SARS-CoV-2, the coronavirus responsible for the COVID-19 pandemic.
Sense is partnering closely with Phillips-Medisize, a Molex company and leading global medical device innovator, developer and manufacturer, to scale-up production of its test in order to meet the growing demand for rapid diagnostics.
Sense’s Veros SARS-CoV-2 is a simple disposable test that uses a nasal swab sample to give an ultra-rapid result without the need for any instrumentation. As a molecular test its performance is equivalent to Gold Standard laboratory tests but it is easy to use in any setting and results are available in under 10 minutes. The test is fully self-contained and can be widely distributed to wherever it is needed, overcoming the logistical and contamination problems associated with machine-based testing.