Globe Newswire04.02.20
Circadia Health is pledging $3 million in medical devices and services towards global COVID-19 relief efforts. The company has expedited the release of the Contactless Respiratory Monitoring System (C100) to reduce the workload and risk of exposure on frontline health workers in the COVID-19 response. Circadia is committing to an immediate delivery of 2,000 devices and is on track to tripling its manufacturing capacity.
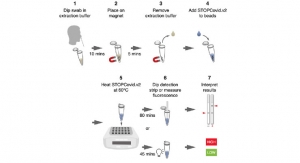
Circadia recognizes the urgent need for remote contactless monitoring of respiratory status at-scale to help: triage patients in-hospital with early detection and mass-monitoring of vulnerable individuals at-home. The Circadia C100 facilitates inexpensive and medical grade respiratory monitoring while reducing patient and healthcare provider contact and exposure to COVID-19. This follows the U.S. Food and Drug Administration’s (FDA) latest guidance to expand the availability and capability of non-invasive remote respiratory monitoring devices.
“The demand for ventilators must be supplemented with the demand for remote monitoring of patients for early detection of respiratory distress. Circadia’s C100 uses AI-powered algorithms and alerts to identify potential worsening of lung function. We have now received exemption from the Malaysian government to ramp up production and are in communication with FDA for expedited review,” said Fares Siddiqui, CEO of Circadia Health.
Circadia has started deploying its AI-powered C100 with a large hospital in Los Angeles and is in ongoing discussions with global governments and health systems.
“Managing and monitoring the influx of patients is overwhelming our global medical systems. Circadia’s proven remote respiratory monitoring of those potentially affected can help prioritize the clinical team’s resources. The need for hands-on review is minimized, with alerts of changes in respiration available if needed. The patients’ progress over time is summarized,” said Chief Medical Advisor Professor Adrian Williams, formerly at Harvard Medical School, UCLA, and former Director of the Lane Fox Respiratory Unit at Guys’ and St Thomas’ Hospital.
Circadia recognizes the urgent need for remote contactless monitoring of respiratory status at-scale to help: triage patients in-hospital with early detection and mass-monitoring of vulnerable individuals at-home. The Circadia C100 facilitates inexpensive and medical grade respiratory monitoring while reducing patient and healthcare provider contact and exposure to COVID-19. This follows the U.S. Food and Drug Administration’s (FDA) latest guidance to expand the availability and capability of non-invasive remote respiratory monitoring devices.
“The demand for ventilators must be supplemented with the demand for remote monitoring of patients for early detection of respiratory distress. Circadia’s C100 uses AI-powered algorithms and alerts to identify potential worsening of lung function. We have now received exemption from the Malaysian government to ramp up production and are in communication with FDA for expedited review,” said Fares Siddiqui, CEO of Circadia Health.
Circadia has started deploying its AI-powered C100 with a large hospital in Los Angeles and is in ongoing discussions with global governments and health systems.
“Managing and monitoring the influx of patients is overwhelming our global medical systems. Circadia’s proven remote respiratory monitoring of those potentially affected can help prioritize the clinical team’s resources. The need for hands-on review is minimized, with alerts of changes in respiration available if needed. The patients’ progress over time is summarized,” said Chief Medical Advisor Professor Adrian Williams, formerly at Harvard Medical School, UCLA, and former Director of the Lane Fox Respiratory Unit at Guys’ and St Thomas’ Hospital.