Business Wire02.14.19
Hologic Inc. announced that its Aptima HIV-1 Quant Dx Assay has received two new CE marks in Europe—for early infant diagnosis (EID) and for testing dried blood spots (DBS). This means the assay can be used to qualitatively detect HIV-1 RNA as an aid in the diagnosis of HIV-1 infected infants under 18 months old, and to test an additional sample type (DBS) to monitor viral load and disease progression in HIV-1 infected individuals in European and African countries. It is the first and only dual-claim assay for both viral load and early infant diagnosis. The dried blood spot claim is particularly important in the African market as it is a much more stable and easily transportable sample type than liquid blood.
The Aptima HIV-1 Quant Dx assay is an in vitro nucleic acid amplification test (NAAT) for the detection and quantitation of HIV type 1 (HIV-1) on the fully automated Panther system. It is intended as an aid in the diagnosis of HIV-1 infection, as a confirmation of HIV-1 infection, and as an aid in the clinical management of patients infected with HIV-1. The Aptima HIV-1 Quant Dx assay may also be used in conjunction with clinical presentation and other laboratory markers for disease prognosis in HIV-1 infected individuals.
“With 25 million people infected with HIV in sub-Saharan Africa alone, there continues to be an urgent need for accessible testing, which is crucial for managing care and reducing the spread of this life-threatening infection,” said João Malagueira, vice president, Europe South and Indirect Markets. “These new product extensions, along with the recent announcement of our Hologic Global Access Initiative, underline Hologic’s commitment to providing accessible testing. They will enable healthcare providers in resource-limited settings to scale up their HIV testing programs to meet the 95-95-95 goals set out by the World Health Organization (WHO).”
The Aptima HIV-1 Quant Dx assay was awarded World Health Organization prequalification for in vitro diagnostics using plasma samples on December 21, 2017. This means that the assay meets WHO standards of quality, safety, performance and reliability, and allows global health organizations to consider the Aptima HIV-1 Quant Dx assay for public sector procurement in resource-limited settings.
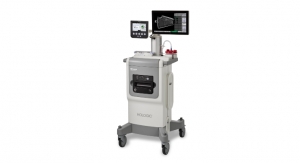
The Aptima HIV-1 Quant Dx assay is processed on Hologic’s Panther system, a market-leading, integrated platform that fully automates molecular testing with true sample-to-result automation, adaptable workflow options, and a broad testing menu. The Panther system is designed to be modular and scalable, accommodating the needs of large, centralized labs as well as smaller, decentralized labs. The Panther system offers the highest throughput per square meter of any comparable molecular diagnostic instrument—up to 320 results in 8 hours in less than one square meter of space.1,2
Hologic has a long-term legacy in the virology space that began two decades ago and spans the development of the first NAATs to screen the blood supply for HIV and HCV (hepatitis C virus). In the early 2000s, Hologic's diagnostic portfolio evolved to include qualitative assays for HIV and HCV, and now also includes quantitative assays for HIV, HBV (hepatitis B virus) and HCV.
References
1 Hologic, Inc. Data on File.
2 Ratnam S, et al. Workflow and maintenance characteristics of five automated laboratory instruments for the diagnosis of sexually transmitted infections. J Clin Microbiol. 2014;52(7); 2299-2304.
The Aptima HIV-1 Quant Dx assay is an in vitro nucleic acid amplification test (NAAT) for the detection and quantitation of HIV type 1 (HIV-1) on the fully automated Panther system. It is intended as an aid in the diagnosis of HIV-1 infection, as a confirmation of HIV-1 infection, and as an aid in the clinical management of patients infected with HIV-1. The Aptima HIV-1 Quant Dx assay may also be used in conjunction with clinical presentation and other laboratory markers for disease prognosis in HIV-1 infected individuals.
“With 25 million people infected with HIV in sub-Saharan Africa alone, there continues to be an urgent need for accessible testing, which is crucial for managing care and reducing the spread of this life-threatening infection,” said João Malagueira, vice president, Europe South and Indirect Markets. “These new product extensions, along with the recent announcement of our Hologic Global Access Initiative, underline Hologic’s commitment to providing accessible testing. They will enable healthcare providers in resource-limited settings to scale up their HIV testing programs to meet the 95-95-95 goals set out by the World Health Organization (WHO).”
The Aptima HIV-1 Quant Dx assay was awarded World Health Organization prequalification for in vitro diagnostics using plasma samples on December 21, 2017. This means that the assay meets WHO standards of quality, safety, performance and reliability, and allows global health organizations to consider the Aptima HIV-1 Quant Dx assay for public sector procurement in resource-limited settings.
The Aptima HIV-1 Quant Dx assay is processed on Hologic’s Panther system, a market-leading, integrated platform that fully automates molecular testing with true sample-to-result automation, adaptable workflow options, and a broad testing menu. The Panther system is designed to be modular and scalable, accommodating the needs of large, centralized labs as well as smaller, decentralized labs. The Panther system offers the highest throughput per square meter of any comparable molecular diagnostic instrument—up to 320 results in 8 hours in less than one square meter of space.1,2
Hologic has a long-term legacy in the virology space that began two decades ago and spans the development of the first NAATs to screen the blood supply for HIV and HCV (hepatitis C virus). In the early 2000s, Hologic's diagnostic portfolio evolved to include qualitative assays for HIV and HCV, and now also includes quantitative assays for HIV, HBV (hepatitis B virus) and HCV.
References
1 Hologic, Inc. Data on File.
2 Ratnam S, et al. Workflow and maintenance characteristics of five automated laboratory instruments for the diagnosis of sexually transmitted infections. J Clin Microbiol. 2014;52(7); 2299-2304.