Globe Newswire08.30.17
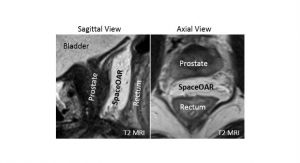
Stereotaxis Inc., a developer of robotic technologies for the treatment of cardiac arrhythmias, announced the results of an independent meta-analysis of studies reporting the comparative safety, efficacy, and efficiency of its Niobe remote magnetic navigation system versus manual navigation in ablation for ventricular tachycardia (VT). The analysis, published in the Journal of Interventional Cardiac Electrophysiology, represents the largest systematic review of VT findings with the Niobe system, comprising data from 779 patients treated at multiple prominent global hospitals.
The results of the analysis demonstrated that the Niobe system is associated with a 39 percent lower risk of VT recurrence (p = 0.003), higher acute procedural success during VT ablation (p = 0.0004) and a 65 percent reduction in risk of complications during VT ablation (p = 0.006). Additionally, procedure time was significantly shorter by an average of 9.8 minutes with the Niobe system (p = 0.04) and fluoroscopy time was significantly reduced by an average of 10.4 minutes (p < 0.00001).
“The Niobe system has overcome many of the limitations inherent with manual ablation techniques in complex procedures, yet studies to date of its impact on VT ablation have been limited in size and consistency. This was an opportunity to pool data for a significant statistical analysis comparing robotic to manual approaches, the results of which verified the Niobe system’s advantages in recurrence rate, procedure time, and complications," said senior author Dr. Dhanunjaya Lakkireddy, professor of cardiovascular medicine at the University of Kansas Medical Center. "We believe the performance benefits lie in the improved reach and precision provided by robotics as well as the continuous consistent tissue contact generated by a magnetic ablation catheter.”
Stereotaxis is currently conducting a prospective, multi-center, randomized control clinical study to demonstrate superiority of robotic cardiac ablation compared to manual approaches in VT patients.
Stereotaxis develops robotic technologies designed to enhance the treatment of arrhythmias and perform endovascular procedures. Its mission is the discovery, development and delivery of robotic systems, instruments, and information solutions for the interventional laboratory. More than 100 issued patents support the Stereotaxis platform. The core components of Stereotaxis’ systems have received regulatory clearance in the United States, European Union, Japan, Canada, China, and elsewhere. The company is based in St. Louis, Mo.
The results of the analysis demonstrated that the Niobe system is associated with a 39 percent lower risk of VT recurrence (p = 0.003), higher acute procedural success during VT ablation (p = 0.0004) and a 65 percent reduction in risk of complications during VT ablation (p = 0.006). Additionally, procedure time was significantly shorter by an average of 9.8 minutes with the Niobe system (p = 0.04) and fluoroscopy time was significantly reduced by an average of 10.4 minutes (p < 0.00001).
“The Niobe system has overcome many of the limitations inherent with manual ablation techniques in complex procedures, yet studies to date of its impact on VT ablation have been limited in size and consistency. This was an opportunity to pool data for a significant statistical analysis comparing robotic to manual approaches, the results of which verified the Niobe system’s advantages in recurrence rate, procedure time, and complications," said senior author Dr. Dhanunjaya Lakkireddy, professor of cardiovascular medicine at the University of Kansas Medical Center. "We believe the performance benefits lie in the improved reach and precision provided by robotics as well as the continuous consistent tissue contact generated by a magnetic ablation catheter.”
Stereotaxis is currently conducting a prospective, multi-center, randomized control clinical study to demonstrate superiority of robotic cardiac ablation compared to manual approaches in VT patients.
Stereotaxis develops robotic technologies designed to enhance the treatment of arrhythmias and perform endovascular procedures. Its mission is the discovery, development and delivery of robotic systems, instruments, and information solutions for the interventional laboratory. More than 100 issued patents support the Stereotaxis platform. The core components of Stereotaxis’ systems have received regulatory clearance in the United States, European Union, Japan, Canada, China, and elsewhere. The company is based in St. Louis, Mo.