08.12.15
Milan, Italy-based Sorin Group, a carduiovascualr device company, received Health Canada approval of its Perceval sutureless aortic valve.
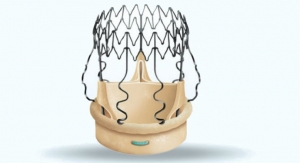
Traditional valves require from 15 to 18 permanent sutures, but the Perceval valve is designed with a self-anchoring frame that enables the surgeon to replace the native diseased valve without sutures.
This innovation reduces procedure time and complexity, leading to shorter stays and fewer complications, according to the company. Additionally, the absence of a suturing ring maximizes the effective orifice area, providing excellent hemodynamics with stable results over time.
The Perceval valve is currently under review by the U.S. Food and Drug Administration. To date, it has been implanted in more than 12,000 patients in over 300 centers, in 34 countries across the world.
“Sorin is committed to innovation that improves patient outcomes, enhances surgical technique and delivers value for hospitals and the health system overall,” said Michel Darnaud, president, Cardiac Surgery Business Unit, Sorin Group. “We believe Perceval meets each of these criteria and has the potential to become the standard of care for patients who require surgical valve replacement procedures. We look forward to working with the surgical community in Canada to bring this important new option to patients.”
“Perceval represents a major advance in surgical aortic valve technology because it reduces aortic cross-clamp times and provides outstanding hemodynamic performance,” said Theodor J.M. Fischlein, M.D., Ph.D., Paracelsus Medical University Cardiovascular Center, Nuremberg, Germany. “A broad range of patient types can benefit from a sutureless approach, including higher-risk patients and those undergoing a concomitant procedure.”
The Canadian approval of Perceval is the latest in a series of milestones for Sorin Group in North America, including the presentation of two major Perceval studies at the 95th Annual American Association of Thoracic Surgery Meeting, the completion of U.S. enrollment for the Perceval investigational device exemption (IDE) trial and the introduction of the Memo 3D ReChord for mitral valve repair and Solo Smart stentless bioprosthesis.
Sorin Group develops, manufactures, and markets medical technologies for cardiac surgery and for the treatment of cardiac rhythm disorders. The company focuses on two major therapeutic areas: Cardiac Surgery (cardiopulmonary products for open heart surgery and heart valve repair or replacement prostheses) and Cardiac Rhythm Management (pacemakers, defibrillators and non invasive monitoring to diagnose arrhythmias and deliver anti-arrhythmia therapies as well as cardiac resynchronization devices for heart failure treatment). The company is in the process of merging with Houston, Texas-based Cyberonics. Once the deal goes through, the combined company will be renamed LivaNova.
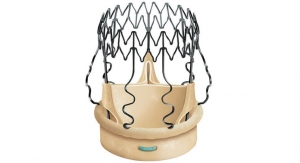
Traditional valves require from 15 to 18 permanent sutures, but the Perceval valve is designed with a self-anchoring frame that enables the surgeon to replace the native diseased valve without sutures.
This innovation reduces procedure time and complexity, leading to shorter stays and fewer complications, according to the company. Additionally, the absence of a suturing ring maximizes the effective orifice area, providing excellent hemodynamics with stable results over time.
The Perceval valve is currently under review by the U.S. Food and Drug Administration. To date, it has been implanted in more than 12,000 patients in over 300 centers, in 34 countries across the world.
“Sorin is committed to innovation that improves patient outcomes, enhances surgical technique and delivers value for hospitals and the health system overall,” said Michel Darnaud, president, Cardiac Surgery Business Unit, Sorin Group. “We believe Perceval meets each of these criteria and has the potential to become the standard of care for patients who require surgical valve replacement procedures. We look forward to working with the surgical community in Canada to bring this important new option to patients.”
“Perceval represents a major advance in surgical aortic valve technology because it reduces aortic cross-clamp times and provides outstanding hemodynamic performance,” said Theodor J.M. Fischlein, M.D., Ph.D., Paracelsus Medical University Cardiovascular Center, Nuremberg, Germany. “A broad range of patient types can benefit from a sutureless approach, including higher-risk patients and those undergoing a concomitant procedure.”
The Canadian approval of Perceval is the latest in a series of milestones for Sorin Group in North America, including the presentation of two major Perceval studies at the 95th Annual American Association of Thoracic Surgery Meeting, the completion of U.S. enrollment for the Perceval investigational device exemption (IDE) trial and the introduction of the Memo 3D ReChord for mitral valve repair and Solo Smart stentless bioprosthesis.
Sorin Group develops, manufactures, and markets medical technologies for cardiac surgery and for the treatment of cardiac rhythm disorders. The company focuses on two major therapeutic areas: Cardiac Surgery (cardiopulmonary products for open heart surgery and heart valve repair or replacement prostheses) and Cardiac Rhythm Management (pacemakers, defibrillators and non invasive monitoring to diagnose arrhythmias and deliver anti-arrhythmia therapies as well as cardiac resynchronization devices for heart failure treatment). The company is in the process of merging with Houston, Texas-based Cyberonics. Once the deal goes through, the combined company will be renamed LivaNova.