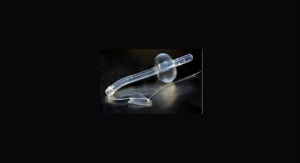
The device features an inflatable balloon that can occlude a variety of arteries up to 4.5 mm in diameter. The catheter regulates the flow of the delivered fluid to maintain a safe pressure, while multiple channels at the distal end of the device help make sure the liquid is distributed well throughout the vessel lumen.
“This technology has two significant scientific advancements," said CEO Nabil Dib M.D., MSc. "The multi-lumen channels provide the smallest luminal diameter available in catheter technology today. The micro channels offer better mixing and dispersion of therapeutic agents, which potentially enhances efficacy."
Additionally, the catheter’s balloon is uniquely designed to minimize pressure on the vessel wall during inflation, conceivably decreasing incidence of vascular trauma compared to conventional technology, Dib noted.
"Since the balloon adapts to different vessel diameters, inventory is reduced, thus decreasing costs” he said. “This technology provides a strategic tool for physicians to deliver better patient care. We are delighted to have gained FDA approval so quickly. TRI’s ability to advance from concept to approval in 31 months is a direct result of our Translational Research infrastructure which is elemental to our ability to expedite scientific discovery for patients.”
TRI is a privately held medical device firm that develops cardiovascular products and treatments.