Business Wire05.24.17
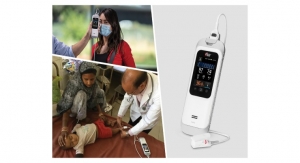
Masimo has introduced Rad-G, a combined pulse oximeter designed primarily for use in pneumonia screening and spot-checking of oxygen saturation (SpO2) in low-resource settings. The development of the device is supported in part by a grant of $4.95 million from the Bill & Melinda Gates Foundation (BMGF), announced in November 2016, as part of a partnership with Masimo to facilitate screening for pneumonia by health workers in low-resource areas.
Rad-G is a low-cost, rugged, handheld pulse oximetry device with a rechargeable battery and LCD display. It uses Masimo Measure-through Motion and Low Perfusion SET pulse oximetry technology to measure SpO2, respiration rate from the Pleth (RRp), pulse rate (PR), and perfusion index (Pi).
Pneumonia remains the single largest treatable infectious cause of death in children worldwide, causing more than 900,000 deaths each year among children under 5 years of age.1 In a study funded by the BMGF Diagnostics Modelling Consortium, the researchers concluded that in settings where supplemental oxygen is available, the addition of pulse oximetry to standard integrated management of child illness protocols could reduce pneumonia mortality rates.2 More recently, the World Health Organization (WHO) has been conducting a multi-country evaluation of enhanced community case management of pneumonia with the use of Masimo SET pulse oximetry by community health workers.3
Enhancing patient screening is critically important to reducing the global burden of pneumonia. Moreover, enhanced screening may empower healthcare providers by supporting informed decisions related to pneumonia diagnosis and treatment, with the appropriate administration of antibiotics and oxygen therapy when needed. Masimo and BMGF hope that Masimo pulse oximetry technology, already used to monitor approximately 100 million patients around the world, can help better screen children in even the most challenging conditions.
“The introduction of the Rad-G is a critical milestone in our partnership with the Bill & Melinda Gates Foundation to help improve pneumonia screening. We are grateful to have the opportunity to bring our proven SET pulse oximetry technology to areas of the world that are in desperate need of better healthcare, and look forward to making a positive difference in the lives of many children,” said Joe Kiani, founder and CEO of Masimo.
Rad-G is currently not available for sale in the United States, Canada, or the European Union.
References
1. Pneumonia Fact Sheet, World Health Organization (WHO), September 2016. http://www.who.int/mediacentre/factsheefts/fs331/en/.
2. Floyd J et al. Evaluating the impact of pulse oximetry on childhood pneumonia mortality in resource-poor settings. Nature. 2015 Dec 3;528(7580):S53-9.
3. World Health Organization (WHO), 2016.
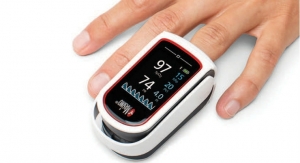
Masimo develops innovative noninvasive monitoring technologies. In 1995, the company debuted Masimo SET Measure-through Motion and Low Perfusion pulse oximetry, which has been shown in multiple studies to significantly reduce false alarms and accurately monitor for true alarms. Masimo SET has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,1 improve CCHD screening in newborns,2 and, when used for continuous monitoring with Masimo Patient SafetyNet in post-surgical wards, reduce rapid response activations and costs.3,4,5 Masimo SET is estimated to be used on more than 100 million patients in leading hospitals and other healthcare settings around the world,6 including 17 of the top 20 hospitals listed in the 2016-17 U.S. News and World Report Best Hospitals Honor Roll.7 In 2005, Masimo introduced rainbow Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb), oxygen content (SpOC), carboxyhemoglobin (SpCO), methemoglobin (SpMet), and more recently, Pleth Variability Index (PVi) and Oxygen Reserve Index (ORi), in addition to SpO2, pulse rate, and perfusion index (PI). In 2014, Masimo introduced Root, an intuitive patient monitoring and connectivity platform with the Masimo Open Connect (MOC-9) interface, enabling other companies to augment Root with new features and measurement capabilities. Masimo is also taking an active leadership role in mHealth with products such as the Radius-7 wearable patient monitor, iSpO2 pulse oximeter for smartphones, and the MightySat fingertip pulse oximeter.
References
1. Castillo A et al. Prevention of Retinopathy of Prematurity in Preterm Infants through Changes in Clinical Practice and SpO2 Technology. Acta Paediatr. 2011 Feb;100(2):188-92.
2. de-Wahl Granelli A et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ. 2009;338.
3. Taenzer AH et al. Impact of Pulse Oximetry Surveillance on Rescue Events and Intensive Care Unit Transfers: A Before-And-After Concurrence Study. Anesthesiology. 2010; 112(2):282-287.
4. Taenzer AH et al. Postoperative Monitoring – The Dartmouth Experience. Anesthesia Patient Safety Foundation Newsletter. Spring-Summer 2012.
5. McGrath SP et al. Surveillance Monitoring Management for General Care Units: Strategy, Design, and Implementation. The Joint Commission Journal on Quality and Patient Safety. 2016 Jul;42(7):293-302.
6. Estimate: Masimo data on file.
7. http://health.usnews.com/health-care/best-hospitals/articles/best-hospitals-honor-roll-and-overview.
Rad-G is a low-cost, rugged, handheld pulse oximetry device with a rechargeable battery and LCD display. It uses Masimo Measure-through Motion and Low Perfusion SET pulse oximetry technology to measure SpO2, respiration rate from the Pleth (RRp), pulse rate (PR), and perfusion index (Pi).
Pneumonia remains the single largest treatable infectious cause of death in children worldwide, causing more than 900,000 deaths each year among children under 5 years of age.1 In a study funded by the BMGF Diagnostics Modelling Consortium, the researchers concluded that in settings where supplemental oxygen is available, the addition of pulse oximetry to standard integrated management of child illness protocols could reduce pneumonia mortality rates.2 More recently, the World Health Organization (WHO) has been conducting a multi-country evaluation of enhanced community case management of pneumonia with the use of Masimo SET pulse oximetry by community health workers.3
Enhancing patient screening is critically important to reducing the global burden of pneumonia. Moreover, enhanced screening may empower healthcare providers by supporting informed decisions related to pneumonia diagnosis and treatment, with the appropriate administration of antibiotics and oxygen therapy when needed. Masimo and BMGF hope that Masimo pulse oximetry technology, already used to monitor approximately 100 million patients around the world, can help better screen children in even the most challenging conditions.
“The introduction of the Rad-G is a critical milestone in our partnership with the Bill & Melinda Gates Foundation to help improve pneumonia screening. We are grateful to have the opportunity to bring our proven SET pulse oximetry technology to areas of the world that are in desperate need of better healthcare, and look forward to making a positive difference in the lives of many children,” said Joe Kiani, founder and CEO of Masimo.
Rad-G is currently not available for sale in the United States, Canada, or the European Union.
References
1. Pneumonia Fact Sheet, World Health Organization (WHO), September 2016. http://www.who.int/mediacentre/factsheefts/fs331/en/.
2. Floyd J et al. Evaluating the impact of pulse oximetry on childhood pneumonia mortality in resource-poor settings. Nature. 2015 Dec 3;528(7580):S53-9.
3. World Health Organization (WHO), 2016.
Masimo develops innovative noninvasive monitoring technologies. In 1995, the company debuted Masimo SET Measure-through Motion and Low Perfusion pulse oximetry, which has been shown in multiple studies to significantly reduce false alarms and accurately monitor for true alarms. Masimo SET has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,1 improve CCHD screening in newborns,2 and, when used for continuous monitoring with Masimo Patient SafetyNet in post-surgical wards, reduce rapid response activations and costs.3,4,5 Masimo SET is estimated to be used on more than 100 million patients in leading hospitals and other healthcare settings around the world,6 including 17 of the top 20 hospitals listed in the 2016-17 U.S. News and World Report Best Hospitals Honor Roll.7 In 2005, Masimo introduced rainbow Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb), oxygen content (SpOC), carboxyhemoglobin (SpCO), methemoglobin (SpMet), and more recently, Pleth Variability Index (PVi) and Oxygen Reserve Index (ORi), in addition to SpO2, pulse rate, and perfusion index (PI). In 2014, Masimo introduced Root, an intuitive patient monitoring and connectivity platform with the Masimo Open Connect (MOC-9) interface, enabling other companies to augment Root with new features and measurement capabilities. Masimo is also taking an active leadership role in mHealth with products such as the Radius-7 wearable patient monitor, iSpO2 pulse oximeter for smartphones, and the MightySat fingertip pulse oximeter.
References
1. Castillo A et al. Prevention of Retinopathy of Prematurity in Preterm Infants through Changes in Clinical Practice and SpO2 Technology. Acta Paediatr. 2011 Feb;100(2):188-92.
2. de-Wahl Granelli A et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ. 2009;338.
3. Taenzer AH et al. Impact of Pulse Oximetry Surveillance on Rescue Events and Intensive Care Unit Transfers: A Before-And-After Concurrence Study. Anesthesiology. 2010; 112(2):282-287.
4. Taenzer AH et al. Postoperative Monitoring – The Dartmouth Experience. Anesthesia Patient Safety Foundation Newsletter. Spring-Summer 2012.
5. McGrath SP et al. Surveillance Monitoring Management for General Care Units: Strategy, Design, and Implementation. The Joint Commission Journal on Quality and Patient Safety. 2016 Jul;42(7):293-302.
6. Estimate: Masimo data on file.
7. http://health.usnews.com/health-care/best-hospitals/articles/best-hospitals-honor-roll-and-overview.