12.19.13
Philadelphia, Pa.-based Echo Therapeutics has forged a strategic collaboration agreement with Medical Technologies Innovation Asia Ltd. (MTIA), Hong Kong, for a license arrangement and equity investment in Echo. According to the terms, Echo grants MTIA rights to develop, manufacture, market and distribute Echo’s Symphony CGM system on an exclusive basis for the Chinese market, including the Peoples’ Republic of China, Hong Kong, Macau and Taiwan.
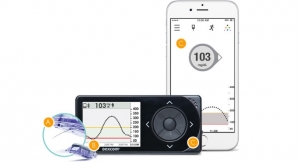
Symphony CGM is touted as a non-invasive (needle-free), wireless, continuous glucose monitoring system designed to provide accurate, real-time blood glucose data conveniently and continuously.
Under the terms of the Agreement, MTIA is responsible for development costs, as well as manufacturing and marketing costs, relating to the Symphony CGM system for the Chinese market, and for obtaining regulatory approval for the product in the licensed territories. Upon regulatory approval from the China Food and Drug Administration or upon Echo’s termination of the agreement, MTIA is entitled to reimbursement of development costs up to $1.5 million in the form of shares of Echo’s common stock valued at $2.71, which stock was valued at on Dec. 9, 2013, the day the deal was signed. The parties will share net sales of the product generated in the territory. The agreement is good for 10 years, subject to certain rights to earlier termination for breach, change of control and certain performance obligations.
The worldwide market for glucose monitoring devices is approximately $10 billion, and is largely composed of blood glucose meters and test strips which provide single blood glucose values. According to Echo, Symphony has the potential to offer a non-invasive, continuous glucose monitoring solution for use in the emerging hospital market. This strategic partnership offers Echo both the potential to accelerate Symphony’s commercialization in China and to access to the Chinese market, where MTIA has established sales channels in more than 1,000 hospitals spanning the country’s provinces.
“This collaboration addresses a specific market need in China and we believe that, with our medical device expertise and extensive sales channels, the Symphony CGM System has the potential to revolutionize the standard of care for glucose monitoring in China,” said Bai Ge, managing director of MTIA. “We are exceptionally pleased to work together with Echo and look forward to a long and productive partnership.”
As part of the agreement, MTIA and its affiliates purchases Echo stocks amounting to $10 million, giving a boost to the company’s device commercialization efforts.
“We are excited to be working with MTIA in this new relationship to meet the growing glucose monitoring needs in China,” said Robert F. Doman, executive chairman and interim CEO of Echo Therapeutics. “We believe MTIA’s expertise and relationships in China will enhance Echo’s success in this region and our overall value proposition. In addition to strengthening the balance sheet, this $10 million cash infusion, following the recent positive clinical results for use of our Symphony CGM System, represents an important achievement for Echo as we progress toward regulatory clearance.”
Medical Technologies Innovation Asia focuses on the distribution of medical devices for oncology, diabetic, cardiovascular and other aging-related disease.
Echo Therapeutics is in the midst of developing the Symphony CGM system. The company also makes skin-preparation devices for the delivery of pharmaceuticals.
Symphony CGM is touted as a non-invasive (needle-free), wireless, continuous glucose monitoring system designed to provide accurate, real-time blood glucose data conveniently and continuously.
Under the terms of the Agreement, MTIA is responsible for development costs, as well as manufacturing and marketing costs, relating to the Symphony CGM system for the Chinese market, and for obtaining regulatory approval for the product in the licensed territories. Upon regulatory approval from the China Food and Drug Administration or upon Echo’s termination of the agreement, MTIA is entitled to reimbursement of development costs up to $1.5 million in the form of shares of Echo’s common stock valued at $2.71, which stock was valued at on Dec. 9, 2013, the day the deal was signed. The parties will share net sales of the product generated in the territory. The agreement is good for 10 years, subject to certain rights to earlier termination for breach, change of control and certain performance obligations.
The worldwide market for glucose monitoring devices is approximately $10 billion, and is largely composed of blood glucose meters and test strips which provide single blood glucose values. According to Echo, Symphony has the potential to offer a non-invasive, continuous glucose monitoring solution for use in the emerging hospital market. This strategic partnership offers Echo both the potential to accelerate Symphony’s commercialization in China and to access to the Chinese market, where MTIA has established sales channels in more than 1,000 hospitals spanning the country’s provinces.
“This collaboration addresses a specific market need in China and we believe that, with our medical device expertise and extensive sales channels, the Symphony CGM System has the potential to revolutionize the standard of care for glucose monitoring in China,” said Bai Ge, managing director of MTIA. “We are exceptionally pleased to work together with Echo and look forward to a long and productive partnership.”
As part of the agreement, MTIA and its affiliates purchases Echo stocks amounting to $10 million, giving a boost to the company’s device commercialization efforts.
“We are excited to be working with MTIA in this new relationship to meet the growing glucose monitoring needs in China,” said Robert F. Doman, executive chairman and interim CEO of Echo Therapeutics. “We believe MTIA’s expertise and relationships in China will enhance Echo’s success in this region and our overall value proposition. In addition to strengthening the balance sheet, this $10 million cash infusion, following the recent positive clinical results for use of our Symphony CGM System, represents an important achievement for Echo as we progress toward regulatory clearance.”
Medical Technologies Innovation Asia focuses on the distribution of medical devices for oncology, diabetic, cardiovascular and other aging-related disease.
Echo Therapeutics is in the midst of developing the Symphony CGM system. The company also makes skin-preparation devices for the delivery of pharmaceuticals.