Medical Product Outsourcing Magazine
July/August 2021
-

Digitization of the Regulatory Review
The FDA’s roadmap to digitization and remote audits includes more modernized approaches to regulatory oversight.07.22.21
-

Metal Injection Molding and Metal 3D Printing Examination
Both MIM and metal AM serve as alternative options to machining for medtech manufacturers requiring metal components.07.22.21
-
 Regulatory PerspectivesGuidance on Skin Irritation With a Push to Achieve the 3 RsThe publication of ISO 10993-23:2021 marks a paradigm shift in prioritizing in-vitro testing alternatives within the medical device biocompatibility space.07.22.21
Regulatory PerspectivesGuidance on Skin Irritation With a Push to Achieve the 3 RsThe publication of ISO 10993-23:2021 marks a paradigm shift in prioritizing in-vitro testing alternatives within the medical device biocompatibility space.07.22.21
-
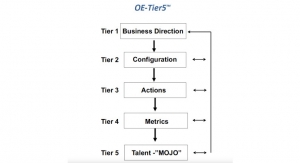 The Supply ChainSupply Chain’s Latest Challenge: The Labor ShortageDuring my career configuring global supply chains, I never imagined a lack of available (or willing) workers would become one of the most formidable challenges.07.22.21
The Supply ChainSupply Chain’s Latest Challenge: The Labor ShortageDuring my career configuring global supply chains, I never imagined a lack of available (or willing) workers would become one of the most formidable challenges.07.22.21
-
 Dollars & SenseTips for Protecting Intellectual Property to Secure FundingThis column provides tips on how to ensure a medtech company’s other valuable intellectual property is ready for due diligence.07.22.21
Dollars & SenseTips for Protecting Intellectual Property to Secure FundingThis column provides tips on how to ensure a medtech company’s other valuable intellectual property is ready for due diligence.07.22.21
-
 Manufacturing MattersCOVID and the U.S.-China Medtech RelationshipThe pandemic has occasioned a drastic realignment of the United States’ leading medtech suppliers.07.22.21
Manufacturing MattersCOVID and the U.S.-China Medtech RelationshipThe pandemic has occasioned a drastic realignment of the United States’ leading medtech suppliers.07.22.21
-
 Best PracticesBack in 3D: AMUG Conference Welcomes Members in the FleshThis was my first visit to AMUG; I spoke about the role of the FDA in regulating medical devices produced via AM.07.22.21
Best PracticesBack in 3D: AMUG Conference Welcomes Members in the FleshThis was my first visit to AMUG; I spoke about the role of the FDA in regulating medical devices produced via AM.07.22.21
-
 EditorialThoughts and Observations from the 2021 Top Companies ReportsThe Top Companies issue is a fantastic way to catch up with what's happening at the leading firms, but COVID dominated this year's reporting.07.22.21
EditorialThoughts and Observations from the 2021 Top Companies ReportsThe Top Companies issue is a fantastic way to catch up with what's happening at the leading firms, but COVID dominated this year's reporting.07.22.21
-
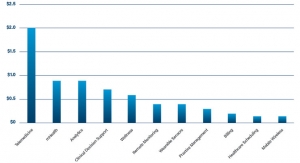 DatawatchDigital Heath Funding Is on FireDigital health funding nearly doubled in Q1 2021 to almost $7.2 billion and 179 deals as compared to Q4 2020.07.22.21
DatawatchDigital Heath Funding Is on FireDigital health funding nearly doubled in Q1 2021 to almost $7.2 billion and 179 deals as compared to Q4 2020.07.22.21
-
 Top30The 2021 MPO Top 30 Medical Device Companies ReportThe COVID-19 pandemic struck the medical device industry more than most.07.22.21
Top30The 2021 MPO Top 30 Medical Device Companies ReportThe COVID-19 pandemic struck the medical device industry more than most.07.22.21
-
 ColumnsThe Top 30 and Their Impact on the C-SuiteMPO’s top companies report is an excellent time to reflect upon the wide range of impacts these industry giants have on the medical device industry.07.22.21
ColumnsThe Top 30 and Their Impact on the C-SuiteMPO’s top companies report is an excellent time to reflect upon the wide range of impacts these industry giants have on the medical device industry.07.22.21
-
 ColumnsRevolutionizing the Complaint Management Process with Artificial IntelligenceDigital technology and artificial intelligence (AI) are changing the way organizations manage quality processes.07.22.21
ColumnsRevolutionizing the Complaint Management Process with Artificial IntelligenceDigital technology and artificial intelligence (AI) are changing the way organizations manage quality processes.07.22.21
















