Sam Brusco, Associate Editor10.16.18
The MedTech Conference (powered by AdvaMed) is one of my favorite events to attend. Hosted at the Pennsylvania Convention Center in Philadelphia this year, the four-day conference features panel sessions with industry veterans, showcases of innovative technologies, entertaining plenary speakers, and ample networking opportunities, among many other useful and informative events. It’s always difficult to select a highlight with so much medtech material offered.
This year, the “super session” panel “Linking Military Service and Innovation: Perspectives from Medical Devices Veterans” captured my attention. Each panelist had served several years in the military:
Such concerns were the breeding ground for MVPvets, a collaborative endeavor between Abiomed and AdvaMed that began in 2012 to address the challenges of life science talent demands and job needs for qualified transitioning veterans, headed by Minogue. In 2014, MVPvets became a 501(c)3 not-for-profit national endeavor, with the mission of re-careering 5,000 veterans into the life sciences by 2020.
MVPvets offers veterans access to thousands of job postings and personalized matching each month, a national mentor platform, career exploration, company-hosted re-careering events, and local and regional networking, among other benefits. It serves to bring veterans, mentors, and employers together to support the science, technology, engineering, mathematics, or business functions of life sciences organizations, providing veterans a seamless transition to the industry.
The main reason the medical technology industry is so attractive to veterans, each panelist agreed, was the sense of meaning and unity coupled with a shared goal for the greater good, which is common to both the military and medtech. Each is concerned with saving lives, neither set of missions can be accomplished without a team’s support, and the career becomes so much more than “work.”
“Military vets flock to medtech because it’s 24/7, and there’s the same sense of purpose,” Minogue said. Herrera echoed the sentiment, saying, “I needed to find the same sense of unity, and found the same meaning and impact in medtech.”
Herrera’s story of founding Spinal Singularity arose from a serious injury he sustained during his second tour of Afghanistan in 2012. While he led a 20-man patrol in the country’s Helmand Province, his Marines were ambushed. While stranded on the roof of a building with his men, a Taliban sniper shot Herrera through his shoulder. The bullet lodged between his C6 and C7 vertebrae, rendering him paraplegic. He continued to give orders during the firefight, and his team was eventually safely evacuated.
Rather than resigning himself to his fate, Herrera learned as much about his spinal cord injury as possible after leaving the Marines. One of his largest problems—which many spinal cord injury patients experience—was bladder management. (Currently, patients use disposable catheters several times a day to empty their bladder.) Just three years after his injury, Herrera graduated from UCLA business school’s executive MBA program, and launched Spinal Singularity soon thereafter.
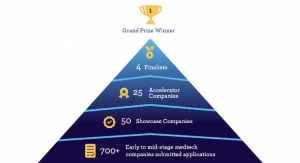
Spinal Singularity’s main project is the Connected Catheter, a fully internal, extended-use, smart catheter system intended to replace single-use catheters. The goal, Herrera said, was to allow spinal cord injury patients to catheterize themselves once a month instead of several times a day. The company has already attained several awards—including semi-finalist status in the MedTech Innovator Accelerator program. “Mike and Alex were a big help [there],” Herrera recalled.
More so than its individual projects, the company seeks to transform the way medical devices are developed. Rather than traditionally cumbersome, time-consuming, and expensive traditional industry research, Spinal Singularity aims to create a product accountable to the public, and developed with feedback from doctors, engineers, and potential users.
The Connected Catheter is currently in clinical studies—its first patient was treated in March. In late August, Spinal Singularity received a grant from the Triumph Foundation that it intends to use to support upcoming clinical trials. The company also added a number of sites to its current feasibility trial, including Los Angeles, Phoenix, Minneapolis, and Mt. Laurel, N.J. Upon collection of the data, the company plans to file for CE mark approval and 510(k) clearance, and Herrera hopes to launch the Connected Catheter next year.
This year, the “super session” panel “Linking Military Service and Innovation: Perspectives from Medical Devices Veterans” captured my attention. Each panelist had served several years in the military:
- Alex Gorsky, chairman and CEO of Johnson & Johnson—former U.S. Army ranger, finishing his career as captain
- Michael Minogue, chairman, president, and CEO of Abiomed—former U.S. Army Infantry officer who served in Desert Storm and earned a Bronze Star
- Derek Herrera, founder and chief technology officer of Spinal Singularity—former Marine Corp. captain who did tours in Iraq and Afghanistan following the Sept. 11 terrorist attacks and earned a Bronze Star
Such concerns were the breeding ground for MVPvets, a collaborative endeavor between Abiomed and AdvaMed that began in 2012 to address the challenges of life science talent demands and job needs for qualified transitioning veterans, headed by Minogue. In 2014, MVPvets became a 501(c)3 not-for-profit national endeavor, with the mission of re-careering 5,000 veterans into the life sciences by 2020.
MVPvets offers veterans access to thousands of job postings and personalized matching each month, a national mentor platform, career exploration, company-hosted re-careering events, and local and regional networking, among other benefits. It serves to bring veterans, mentors, and employers together to support the science, technology, engineering, mathematics, or business functions of life sciences organizations, providing veterans a seamless transition to the industry.
The main reason the medical technology industry is so attractive to veterans, each panelist agreed, was the sense of meaning and unity coupled with a shared goal for the greater good, which is common to both the military and medtech. Each is concerned with saving lives, neither set of missions can be accomplished without a team’s support, and the career becomes so much more than “work.”
“Military vets flock to medtech because it’s 24/7, and there’s the same sense of purpose,” Minogue said. Herrera echoed the sentiment, saying, “I needed to find the same sense of unity, and found the same meaning and impact in medtech.”
Herrera’s story of founding Spinal Singularity arose from a serious injury he sustained during his second tour of Afghanistan in 2012. While he led a 20-man patrol in the country’s Helmand Province, his Marines were ambushed. While stranded on the roof of a building with his men, a Taliban sniper shot Herrera through his shoulder. The bullet lodged between his C6 and C7 vertebrae, rendering him paraplegic. He continued to give orders during the firefight, and his team was eventually safely evacuated.
Rather than resigning himself to his fate, Herrera learned as much about his spinal cord injury as possible after leaving the Marines. One of his largest problems—which many spinal cord injury patients experience—was bladder management. (Currently, patients use disposable catheters several times a day to empty their bladder.) Just three years after his injury, Herrera graduated from UCLA business school’s executive MBA program, and launched Spinal Singularity soon thereafter.
Spinal Singularity’s main project is the Connected Catheter, a fully internal, extended-use, smart catheter system intended to replace single-use catheters. The goal, Herrera said, was to allow spinal cord injury patients to catheterize themselves once a month instead of several times a day. The company has already attained several awards—including semi-finalist status in the MedTech Innovator Accelerator program. “Mike and Alex were a big help [there],” Herrera recalled.
More so than its individual projects, the company seeks to transform the way medical devices are developed. Rather than traditionally cumbersome, time-consuming, and expensive traditional industry research, Spinal Singularity aims to create a product accountable to the public, and developed with feedback from doctors, engineers, and potential users.
The Connected Catheter is currently in clinical studies—its first patient was treated in March. In late August, Spinal Singularity received a grant from the Triumph Foundation that it intends to use to support upcoming clinical trials. The company also added a number of sites to its current feasibility trial, including Los Angeles, Phoenix, Minneapolis, and Mt. Laurel, N.J. Upon collection of the data, the company plans to file for CE mark approval and 510(k) clearance, and Herrera hopes to launch the Connected Catheter next year.