Michael Barbella, Managing Editor09.01.17
There never seemed to be enough hours in the day for Bill Thompson.
The Lexington, Ky., octogenarian liked to stay busy—raking, fixing, sweeping, straightening, organizing—anything really, as long as it kept him occupied and fairly active. He usually lined up more projects in a day than time permitted, and never seemed lacking for new pursuits.
It was a simple yet fulfilling existence, marred only by the intrusion of a massive stroke not too long ago.
Thompson’s wife Donna was home when he suddenly collapsed on the bathroom floor one morning after coffee. She immediately recognized the culprit that had robbed her beloved husband of his speech and mobility. “His voice was slurred, he could not respond to me, and he couldn’t move,” she recalled.
Paramedics arrived within minutes and rushed Bill to the Baptist Health Hospital, where scans revealed a blood clot in his brain. He received the clot-busting drug tPA (tissue plasminogen activator) intravenously, but the blockage was too big to be dissolved quickly by the medication. Doctors instead used a stent retrieval device to snag the blockage, surgically inserting the self-expanding mesh mechanism into Thompson’s femoral artery and guiding it carefully into his brain to latch onto the gel-like clumps of blood interfering with his cognitive functions. Within minutes of the clot’s removal, Bill was talking and moving normally again.
“It’s something,” Bill said, still amazed at the technology. “I don’t see how they could go up through that artery and get all the way up to the brain, past the heart, and take that clot out.”
Indeed, clot removal through stent retrieval seems almost magical: The minimally invasive procedure takes as little as an hour to perform and can significantly reduce the risk of irreversible brain damage. Studies have shown that one in three patients undergoing thrombectomy (stent retrieval) regain full mobility after their strokes, compared to one in five using conventional methods.
Thompson signed on to the former group, returning to his daily chores in just a few weeks. But his recovery was not yet complete.
Doctors had classified his stroke as “cryptogenic,” meaning there was no known cause. Heart arrhythmias are often responsible, but their sporadic nature (with months passing between episodes) makes them difficult to detect by traditional monitoring methods. Without a firm diagnosis and proper treatment, Thompson’s risk for future strokes would likely increase five-fold.
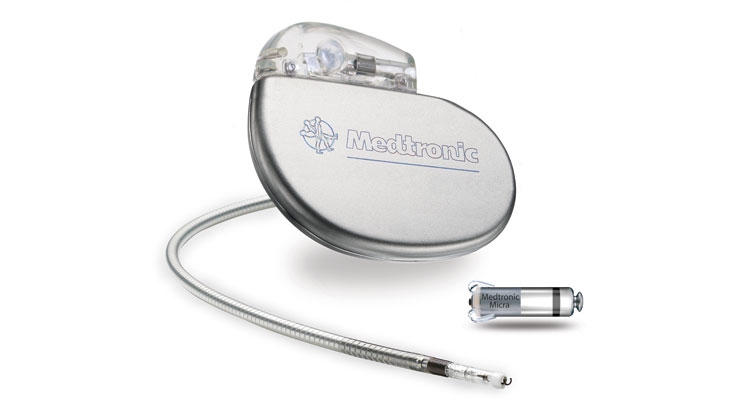
To reduce that risk, Baptist Health Hospital cardiologists implanted a miniaturized heart monitor called Reveal LINQ under Thompson’s skin. Developed by Medtronic plc and approved by U.S. regulators in 2014, the device is 1 cubic centimeter (about one-third the size of a triple-A battery) and designed to keep a watchful eye on cardiac rhythms for up to three years continuously. It also uses wireless telemetry to remotely record the heart’s electrical activity.
Reveal LINQ eventually proved its worth in Thompson’s case, detecting atrial fibrillation in eight months (only slightly longer than the 182-day median recorded in clinical trials). Doctors are treating the condition with blood thinners, enabling Thompson to spend quality time with both his family and his projects.
Reveal LINQ is part of a growing class of incredibly shrinking medical devices that are providing real benefits for patients. Driven by skyrocketing healthcare costs, aging populations, chronic disease, and shorter hospitalizations, implantable devices are being developed in sizes never before thought possible. Medtronic’s Micra Transcatheter Pacing System, for example, is only an inch in length and attaches to the heart through small tines. Leadless and featuring a 12-year estimated battery life, the Micra—touted as the “world’s smallest pacemaker”—has a 99.2 percent implant success rate and 48 percent fewer complications than other, larger products.
“We are looking at the beginning of the future,” said John Hummel, M.D., a Medtronic consultant and professor of cardiovascular medicine at Ohio State University. “We will no longer pace the heart the way we have in the last 20 to 30 years. This is fundamentally a paradigm shift in how we’ll deliver this therapy.”
The catalyst for this shift lies in the technology developed in other industries and modified for healthcare—specifically, the chips and sensors offering more functions and greater processing capacity to cell phones and computers. The latest generations of sensors and microprocessors are smaller, more robust, and more sensitive than their predecessors, and thus are spearheading new possibilities for implantable medical devices.
Miniaturization is key to developing implantable products that have a minimal adverse impact on quality of life. Shrinking devices to a fraction of their former size also has improved power efficiency and circuit operating speed by reducing the resistive and reactive effects of traditional wiring and integrated circuit lead systems.
“Most of the progress in miniaturizing implantable medical devices over the last 20 years has been achieved through a combination of integrated circuits, surface-mount devices, and specialized printed circuit board design,” notes BCC Research analyst Andrew McWilliams. “Breakthroughs in MEMS (microelectromechanical systems) manufacturing should lead to smaller implants with greatly increased capabilities. There is every reason to believe that pacemakers and other implantable devices will continue to get smaller and more sophisticated over time.”
All while generating some mighty big profits: Miniaturization is one of the major growth drivers of the global implantable medical device market, according to industry data. Markets and Markets Research projects the sector to expand 7.8 percent annually over the next five years to reach $26.75 billion by 2022.
The medical implantables market comprises a large assortment of healthcare technologies to achieve specific therapeutic solutions; they can range from orthopedic implants, electronic devices, or drug-eluting scaffolds to artificial body parts and/or organs. These technologies, however, are accompanied by an array of development obstacles that can hinder efforts to design a device that effectively treats diseases or conditions. Among the challenges associated with developing a medical implant are power concerns, data and device security, communications protocols, and of course, size.
To better understand the factors and concerns affecting implant development, Medical Product Outsourcing spoke to nearly a half-dozen device manufacturing professionals and consultants over the last few weeks. Participants included:
Julie Cameron, vice president of sales and marketing, Biomaterials; Jim Lambert, director of life sciences technology and innovation, Biomaterials; and Jesse Gregory, director of technology and innovation, Trelyst, for Avantor, a global supplier of ultra-high purity materials for the life sciences and advanced technology markets. The company—which merged with NuSil Technology LLC last spring—provides performance materials and solutions for the production and research needs of customers in the biotechnology, pharmaceutical, medical device, diagnostics, aerospace and defense, and semiconductor industries.
Kenneth A. Fine, president and co-founder of Mansfield, Mass.-based Proven Process Medical Devices Inc., a 23-year-old designer and manufacturer of Class II and Class III healthcare products.
Brian Highley, CEO of Cirtec Medical, an outsource partner for product design, development, and manufacturing of Class II and Class III medical devices. The company is based in Brooklyn Park, Minn.
Michael Barbella: What factors are driving growth in the implantable medical device market? Are these factors expected to continue or change in the coming years?
Julie Cameron, Jesse Gregory, Jim Lambert: The changing demographics—increased disposable income and aging population, globally—have driven growth. Our Trelyst business is addressing the trend toward combination devices, which provide increased compliance, and targeted steady dosing with fewer side effects. While we can’t disclose confidential projects, we are seeing two trends: one to de-risk procedures and another around ability for sensing. Both of these have created unique material needs where NuSil’s ability and willingness to customize has created novel solutions. These factors are expected to continue to grow in the coming years.
Ken Fine: Based on what our customers are saying, for the foreseeable future, the demand for implanted medical devices will continue to grow. The factors driving the growth include:
Brian Highley: The growth of active implantables is being driven by innovation in the end markets that we’re servicing, primarily neuromodulation. One of the reasons we’re so focused on the neuromodulation market is the size—this is a $3.5 billion market growing at 11 to 12 percent, and a lot of that is due to all the new therapies that are benefitting from neuromodulation. Why neuromodulation? We’re finding that stimulating the nerves can mitigate many of the symptoms associated with diseases such as chronic inflammation, hypertension, and obesity. Traditionally, neurostimulation devices have targeted pain management, which is the largest market. Treating tremors for Parkinson’s patients is another application that has been available for many years. Today there are a vast number of neurostimulation companies studying the benefits of stimulation therapy to treat diabetes, obesity, incontinence, chronic inflammation, chronic hypertension, stroke, PTSD, and other conditions traditionally treated with pharmaceuticals. There’s a lot of innovation in this space, driving the growth rates that I mentioned previously.
I believe the growth rates have been understated as they do not fully capture the trend we’re seeing of non-traditional entities entering the neuromodulation space. This is accomplished through collaborative agreements and spin-off organizations designed to accelerate the speed of innovation while not being burdened by the complex product development process that typically exists within a large medical device OEM. For example, a couple of years ago, GlaxoSmithKline (GSK) stepped into the market, and recently partnered with Verily Life Sciences to create Galvani Bioelectronics. They have committed $715 million over seven years to explore neuromodulation. We know there are other large pharma companies that are beginning to explore this area, as well as other large medical device OEMs. I personally believe we’re going to see more growth than what’s being projected over the next decade.
Barbella: What are the most challenging aspects of designing/developing an implantable medical device?
Cameron, Gregory, Lambert: Regulatory approvals tend to be the most challenging aspect of commercialization.
Fine: There are a number of challenging aspects to developing an implantable medical device.
Highley: Typically it’s the regulatory approval process. I believe these devices are relatively straightforward—we know them very well, they’re characterized by the design and the construction. It’s just that the regulatory approval process is pretty extensive and quite expensive. There’s a significant amount of testing and clinical data that has to be collected to get approval for a product. Many companies will go to Europe to launch their product first. This was a trend discussed when I was over on the other side of the business—the traditional Class II device business. But many of these Class III devices start off in Europe because the regulatory process is a bit easier and actually helps them work toward FDA approval, which can take years and a significant financial investment. It is not uncommon to spend over one hundred million dollars to get a product developed and launched in the United States. It’s an expensive endeavor.
Barbella: How have you addressed these challenges?
Cameron, Gregory, Lambert: Our team stays current with evolving regulations globally to support our materials in our customers’ markets. NuSil has more than 20 years’ of experience providing MAF and DMF supported long-term implantable materials to provide our customers’ path for approval.
Fine: Our company has a well seasoned medical device engineering staff that has been working on implanted medical devices for many years. They work within an environment that has a robust quality system to ensure that the activities meet regulatory and quality standards throughout the world. We work with companies, universities, and medical institutions around the world applying our engineering skills, and gaining exposure and additional expertise with a wide variety of implanted and external medical device technologies.
Barbella: How are implantable device designers and/or manufacturers addressing the demand for miniaturization and ever-smaller products? Is there a limit to how small an implantable device can be?
Cameron, Gregory, Lambert: Who would have thought that pacemakers, which were initially the size of a hockey puck when first introduced, would ever be the size of a pill—but they have. Technology in microelectronics, semiconductors, and microfluidics will contribute to increased miniaturization. We don’t think we’ve reached the limit to how small. There are certainly constraints in physics.
Fine: At some point there is a limit to how small an implantable device can be by the nature of what it is trying to accomplish and how it needs to interface with the body to accomplish its function. For active implantable devices, the size may be limited by the amount of energy required for the device to function over several years. Battery technology and density has improved to allow for smaller batteries. Also, wireless energy transfer systems have allowed for smaller rechargeable batteries or devices that do not require an internal primary battery. In addition, chip level circuit integration and novel electronics packaging have allowed engineers to develop smaller active implantable devices. Other implantable devices may have particular strength or robustness requirements, such as with orthopedic implants. As materials improve, the mass of these devices can be reduced.
Highley: A lot of the innovation going on in this space today is miniaturization. Unfortunately, the market tends to move at a glacial pace because change is difficult to implement and there’s a lot of technology that has to be developed. The way to think about miniaturization, I believe, is to get one of these neurostimulators to a point where you can implant it minimally invasively. When that happens the market should expand rapidly. Take for example, a traditional-sized neurostimulator that’s been developed to treat diabetes. Most people would say, “I don’t want this large implant in me to treat my diabetes, I’m going to treat it with medication.” But if I can get the implant small enough to implant minimally invasively, most people will say, “Yes, now that is a viable option for me.” So ideally, we’re working toward these miniaturized, minimally invasively implantable devices that I believe will expand the market rapidly.
Miniaturization is not just in neuromodulation, it can be expanded to other markets as well, including ventricular assist devices. Of course, these aren’t going to get to the size that is minimally invasively implantable like a nerve stimulator, but Cirtec is developing fully implantable ventricular assist devices, meaning there’s no percutaneous line sticking out of your abdomen tying it to an external battery pack. The battery is fairly small and implanted internally. We believe when you’re able to miniaturize these devices, implant them less invasively and have them fully implanted, eliminating the external battery pack, that market will expand as well. So miniaturization is driving a lot of innovation in the active implantable space.
Of course there’s a limit to how small these devices need to be. You need it to be the size of something you can implant minimally invasively through a needle. Let’s use a grain of rice as an example. Is there any reason to go to a size half a grain of rice? Or a third of a grain of rice? Probably not. There’s a point where you may not need to get any smaller for these kinds of devices, but it doesn’t mean people will stop trying.
Barbella: The assimilation of electronics in implantable medical devices has raised hacking concerns within the industry. Is this a legitimate threat? Please explain your answer.
Cameron, Gregory, Lambert: There are implantable devices that function purely on physical/mechanical principles which would have no concern over hacking. For example, hydrocephalic shunts manage function purely on hydraulic pressure.
Fine: Anytime a device, medical or otherwise, can communicate with a network of any type, hacking is a legitimate threat. As more medical devices interact with the internet for data capture or remote programming, robust security measures need to be considered.
Highley: Yes, I think it’s absolutely a legitimate threat but I believe there are, and will be, smart people and companies out there that can add technology to the design to make these devices safe and secure from hacking and other malicious manipulation. Cirtec is working with a few companies that are spearheading security in these implantable devices. This is another great example of the convergence of non-traditional business partners now innovating more and more in the medical device industry; smartphones, apps, wireless communication, security, and privacy will all play a critical role in the future. We can’t move quite as quickly as the consumer world, so in order to include security into a device you have to design it early, develop it, validate it, and obtain all your [regulatory] approvals with it already integrated. Cirtec is working on this now because we know companies will demand it in the future. It is a legitimate threat but it’s slow to implement in this environment.
Barbella: How can implantable medical device developers safeguard their product(s) from unauthorized access, or hackers?
Fine: There are several strategies that can be employed to safeguard medical products from hacking. Medical device developers can leverage existing methodologies such as encryption or multiple level authentication to restrict access to wireless communication. Another strategy may be to implement a totally proprietary communication scheme that does not depend on known standards.
Barbella: Is it reasonable to expect that most—if not all—external medical technologies will eventually become implantable? Why or why not?
Cameron, Gregory, Lambert: There are many external medical devices that are needed for only a short period of time. On the other hand, there are many organs that are transplanted today that we could imagine being replaced with implantable devices.
Fine: I believe that many external technologies will always stay external. Implantable devices are best suited to long-term chronic conditions that cannot be treated easily by other means. Drugs or other means may be a more cost-effective alternative, or present less patient risk than an implanted medical device.
In healthcare, we attempt to either diagnose or treat a particular condition or disease. As some of the non-implantable diagnostic technology improves, it negates the need for an implant. A cardiac loop detector is a good example. There’s a long-term skin patch from LifeWatch that detects arrhythmias, and is replaceable after a few weeks or months. Personally, I’d rather have something on my skin than in my body. Companies also are working on both internal and external versions of glucose monitors.
I think there’s always going to be the yin and yang of technology, and the two are going to fight each other. But at the end of the day, from a use preference point of view, an external device is going to be preferable to an implantable one. Even so, there are some applications where being inside the body is advantageous because the condition or disease can be treated more effectively near the source. As much as implants advance, external devices are going to advance also. At the end of the day, it’s probably more desirable not to cut open the body and put something inside of it.
Barbella: Is it a prerequisite now that implantable medical devices have the ability to communicate wirelessly with consumer electronics like smartphones or tablets? Why or why not?
Cameron, Gregory, Lambert: Having a device that communicates to an external phone or tablet has proven to be useful in some therapies. In addition, it may be useful to detect that a device is present, as well as more information on the device or patient through RFID technology.
Fine: Because there are so many disease states that can be treated by implantable devices, I don’t believe that as a blanket statement medical devices need to communicate with consumer wireless devices. It is likely that many orthopedic implants will remain without a means to communicate outside the body. However, given the advancement of IoT technology, many patients and clinicians involved with implanted medical devices would derive a benefit from such connectivity. For example, implanted diagnostic devices, such as cardiac loop recorders or long term glucose monitors, would benefit from connectivity available to the patient and clinician. Other implanted devices with which a patient or clinician may benefit from interfacing regularly include products such as insulin pumps, pain treatment devices, and a number of neurological treatment devices.
This issue is similar to internal-external (device) argument, whereas one of the best ways to keep something secure is to not connect it to another source. There’s going to be multiple camps. I think what’s going to happen is there will always a camp that believes you have to have the latest bells and whistles, regardless of whether you use them. A lot of the features on early pacemakers were there because the competition had it and not because anybody actually used it. We do a lot of work in implantable drug infusion pumps. Even though constant flow pumps kind of failed and Medtronic pushed programmability, 90 percent of these programmable pumps were set for one rate and left alone. Maybe some of that is you have to have the feature because somebody else has the feature. But it really should be a function of whether or not there is actual value by having the information from the device be available to somebody externally. If that’s the case, then a tablet or going through the cloud is often a good way to do it.
Highley: I believe that’s where these devices are heading, and many Class II devices have been there for years. We see more and more requests for devices that can work with and communicate with other external devices. The problem here is the lifecycle of the external device. For instance, let’s say the obsolescence on an iPhone is 18 months. It’s very tough to design an implantable device that works with an iPhone 6. Two iterations later it’s no longer relevant; you’ll have to continuously update and test your software so that it works with the next-generation phone. I think people have to find a way around doing that all the time. It’s a challenge. People are looking to ensure their devices can communicate with tablets and phones securely but it’s a huge challenge because of the lifecycle of the devices that they’re connecting to, and controlling a potentially lifesaving device is a bit different than updating Candy Crush. The companies that can solve these problems, and frankly, profit from them, are not traditionally aligned to the medical device industry’s regulatory environment. They’re focused on their consumer markets, and it is a steep learning curve to understand the requirements for medical devices and FDA scrutiny. Ultimately, the regulatory bodies, medical device industry leaders, and Silicon Valley experts need to collaborate on this issue. It has started in small pockets of the industry but it will take some time for it to become standardized.
The Lexington, Ky., octogenarian liked to stay busy—raking, fixing, sweeping, straightening, organizing—anything really, as long as it kept him occupied and fairly active. He usually lined up more projects in a day than time permitted, and never seemed lacking for new pursuits.
It was a simple yet fulfilling existence, marred only by the intrusion of a massive stroke not too long ago.
Thompson’s wife Donna was home when he suddenly collapsed on the bathroom floor one morning after coffee. She immediately recognized the culprit that had robbed her beloved husband of his speech and mobility. “His voice was slurred, he could not respond to me, and he couldn’t move,” she recalled.
Paramedics arrived within minutes and rushed Bill to the Baptist Health Hospital, where scans revealed a blood clot in his brain. He received the clot-busting drug tPA (tissue plasminogen activator) intravenously, but the blockage was too big to be dissolved quickly by the medication. Doctors instead used a stent retrieval device to snag the blockage, surgically inserting the self-expanding mesh mechanism into Thompson’s femoral artery and guiding it carefully into his brain to latch onto the gel-like clumps of blood interfering with his cognitive functions. Within minutes of the clot’s removal, Bill was talking and moving normally again.
“It’s something,” Bill said, still amazed at the technology. “I don’t see how they could go up through that artery and get all the way up to the brain, past the heart, and take that clot out.”
Indeed, clot removal through stent retrieval seems almost magical: The minimally invasive procedure takes as little as an hour to perform and can significantly reduce the risk of irreversible brain damage. Studies have shown that one in three patients undergoing thrombectomy (stent retrieval) regain full mobility after their strokes, compared to one in five using conventional methods.
Thompson signed on to the former group, returning to his daily chores in just a few weeks. But his recovery was not yet complete.
Doctors had classified his stroke as “cryptogenic,” meaning there was no known cause. Heart arrhythmias are often responsible, but their sporadic nature (with months passing between episodes) makes them difficult to detect by traditional monitoring methods. Without a firm diagnosis and proper treatment, Thompson’s risk for future strokes would likely increase five-fold.
To reduce that risk, Baptist Health Hospital cardiologists implanted a miniaturized heart monitor called Reveal LINQ under Thompson’s skin. Developed by Medtronic plc and approved by U.S. regulators in 2014, the device is 1 cubic centimeter (about one-third the size of a triple-A battery) and designed to keep a watchful eye on cardiac rhythms for up to three years continuously. It also uses wireless telemetry to remotely record the heart’s electrical activity.
Reveal LINQ eventually proved its worth in Thompson’s case, detecting atrial fibrillation in eight months (only slightly longer than the 182-day median recorded in clinical trials). Doctors are treating the condition with blood thinners, enabling Thompson to spend quality time with both his family and his projects.
Reveal LINQ is part of a growing class of incredibly shrinking medical devices that are providing real benefits for patients. Driven by skyrocketing healthcare costs, aging populations, chronic disease, and shorter hospitalizations, implantable devices are being developed in sizes never before thought possible. Medtronic’s Micra Transcatheter Pacing System, for example, is only an inch in length and attaches to the heart through small tines. Leadless and featuring a 12-year estimated battery life, the Micra—touted as the “world’s smallest pacemaker”—has a 99.2 percent implant success rate and 48 percent fewer complications than other, larger products.
“We are looking at the beginning of the future,” said John Hummel, M.D., a Medtronic consultant and professor of cardiovascular medicine at Ohio State University. “We will no longer pace the heart the way we have in the last 20 to 30 years. This is fundamentally a paradigm shift in how we’ll deliver this therapy.”
The catalyst for this shift lies in the technology developed in other industries and modified for healthcare—specifically, the chips and sensors offering more functions and greater processing capacity to cell phones and computers. The latest generations of sensors and microprocessors are smaller, more robust, and more sensitive than their predecessors, and thus are spearheading new possibilities for implantable medical devices.
Miniaturization is key to developing implantable products that have a minimal adverse impact on quality of life. Shrinking devices to a fraction of their former size also has improved power efficiency and circuit operating speed by reducing the resistive and reactive effects of traditional wiring and integrated circuit lead systems.
“Most of the progress in miniaturizing implantable medical devices over the last 20 years has been achieved through a combination of integrated circuits, surface-mount devices, and specialized printed circuit board design,” notes BCC Research analyst Andrew McWilliams. “Breakthroughs in MEMS (microelectromechanical systems) manufacturing should lead to smaller implants with greatly increased capabilities. There is every reason to believe that pacemakers and other implantable devices will continue to get smaller and more sophisticated over time.”
All while generating some mighty big profits: Miniaturization is one of the major growth drivers of the global implantable medical device market, according to industry data. Markets and Markets Research projects the sector to expand 7.8 percent annually over the next five years to reach $26.75 billion by 2022.
The medical implantables market comprises a large assortment of healthcare technologies to achieve specific therapeutic solutions; they can range from orthopedic implants, electronic devices, or drug-eluting scaffolds to artificial body parts and/or organs. These technologies, however, are accompanied by an array of development obstacles that can hinder efforts to design a device that effectively treats diseases or conditions. Among the challenges associated with developing a medical implant are power concerns, data and device security, communications protocols, and of course, size.
To better understand the factors and concerns affecting implant development, Medical Product Outsourcing spoke to nearly a half-dozen device manufacturing professionals and consultants over the last few weeks. Participants included:
Julie Cameron, vice president of sales and marketing, Biomaterials; Jim Lambert, director of life sciences technology and innovation, Biomaterials; and Jesse Gregory, director of technology and innovation, Trelyst, for Avantor, a global supplier of ultra-high purity materials for the life sciences and advanced technology markets. The company—which merged with NuSil Technology LLC last spring—provides performance materials and solutions for the production and research needs of customers in the biotechnology, pharmaceutical, medical device, diagnostics, aerospace and defense, and semiconductor industries.
Kenneth A. Fine, president and co-founder of Mansfield, Mass.-based Proven Process Medical Devices Inc., a 23-year-old designer and manufacturer of Class II and Class III healthcare products.
Brian Highley, CEO of Cirtec Medical, an outsource partner for product design, development, and manufacturing of Class II and Class III medical devices. The company is based in Brooklyn Park, Minn.
Michael Barbella: What factors are driving growth in the implantable medical device market? Are these factors expected to continue or change in the coming years?
Julie Cameron, Jesse Gregory, Jim Lambert: The changing demographics—increased disposable income and aging population, globally—have driven growth. Our Trelyst business is addressing the trend toward combination devices, which provide increased compliance, and targeted steady dosing with fewer side effects. While we can’t disclose confidential projects, we are seeing two trends: one to de-risk procedures and another around ability for sensing. Both of these have created unique material needs where NuSil’s ability and willingness to customize has created novel solutions. These factors are expected to continue to grow in the coming years.
Ken Fine: Based on what our customers are saying, for the foreseeable future, the demand for implanted medical devices will continue to grow. The factors driving the growth include:
- Demand for solutions to medical problems as the population ages and suffers from chronic diseases.
- More willingness for patients to use technology to solve medical issues. This not only applies to cell phones and tablets and everything around them [patients], but it also applies to medical equipment and devices. People are more willing to accept that technology instead of a drug will provide a solution.
- Availability of new fabrication technologies such as 3D printing that make complex assemblies more cost effective.
- As more products become available, implantable medical devices offer more choices than in the past. Salesmen now have more in their bag related to implantable devices so they can offer hospitals, doctors, and patients more choices, thereby increasing the likelihood that customers will select an implanted device. When you have only a few choices it can be very limiting, but as the options grow, there begins to be an exponential growth of opportunity. I think implanted devices have gone past that tipping point of offering really niche kinds of choices. There are just more choices to be had today. Look at pulse generators—15 to 20 years ago, pulse generators were almost exclusively cardiology devices; they were just starting to cross over into neurology. The current landscape of pulse generators has evolved to where they are being used for deep brain stimulation, chronic pain stimulation, in cardiac, for treating obesity, and treating urinary incontinence. There are just more choices with the technology, and that makes it more acceptable. It also makes it easier for a salesperson to sell something because they’re not a one-trick pony, going in with just one product. They’re going in with a family of devices.
Brian Highley: The growth of active implantables is being driven by innovation in the end markets that we’re servicing, primarily neuromodulation. One of the reasons we’re so focused on the neuromodulation market is the size—this is a $3.5 billion market growing at 11 to 12 percent, and a lot of that is due to all the new therapies that are benefitting from neuromodulation. Why neuromodulation? We’re finding that stimulating the nerves can mitigate many of the symptoms associated with diseases such as chronic inflammation, hypertension, and obesity. Traditionally, neurostimulation devices have targeted pain management, which is the largest market. Treating tremors for Parkinson’s patients is another application that has been available for many years. Today there are a vast number of neurostimulation companies studying the benefits of stimulation therapy to treat diabetes, obesity, incontinence, chronic inflammation, chronic hypertension, stroke, PTSD, and other conditions traditionally treated with pharmaceuticals. There’s a lot of innovation in this space, driving the growth rates that I mentioned previously.
I believe the growth rates have been understated as they do not fully capture the trend we’re seeing of non-traditional entities entering the neuromodulation space. This is accomplished through collaborative agreements and spin-off organizations designed to accelerate the speed of innovation while not being burdened by the complex product development process that typically exists within a large medical device OEM. For example, a couple of years ago, GlaxoSmithKline (GSK) stepped into the market, and recently partnered with Verily Life Sciences to create Galvani Bioelectronics. They have committed $715 million over seven years to explore neuromodulation. We know there are other large pharma companies that are beginning to explore this area, as well as other large medical device OEMs. I personally believe we’re going to see more growth than what’s being projected over the next decade.
Barbella: What are the most challenging aspects of designing/developing an implantable medical device?
Cameron, Gregory, Lambert: Regulatory approvals tend to be the most challenging aspect of commercialization.
Fine: There are a number of challenging aspects to developing an implantable medical device.
- Materials—identifying robust materials that have the needed physical properties as well as biocompatible properties. Materials are always a challenge but I think what’s pushing us is we have some applications that are different than they have been in the past. For example, we are working on ways to deliver biologics internally to a patient or maintain a patient’s biologics (implanting cells and keeping those cells alive until they become active). Some of the innovation associated with these efforts are not compatible with some of the older technologies. Therefore, we have to look at using novel materials to enable these implanted devices to function inside the body. Similarly, because we’re using some materials that were not necessarily used a lot inside the body, finding sterilization options for those materials can be difficult. We are working on a product that generates oxygen inside the body and the materials are not compatible with EtO sterilization or any kind of radiation sterilization, so we’re looking at other sterilization technologies that are considered more novel by the FDA. For traditional devices, there’s not a lot of pressure to push the envelope but there is some pressure with newer applications (biologics) in regards to materials and sterilization. Market and regulatory are the classical challenges that you’ll always see, but some of these technical issues are getting interesting with newer applications.
- Size—establishing the best compromise between the smallest size possible and the size needed to provide good therapy (or diagnostic performance). Other than for certain applications, I generally don’t see a real pressure to push the size envelope. At some point the devices become so small you can’t really handle them all that well anyway.
- Power management—for active implantables, supplying sufficient power to the implant for a reasonable duration within the size constraints of the device.
- Sterilization options—determining is traditional sterilization technologies such as EtO, gamma, e-beam, or steam are suitable, or if a more novel sterilization options is required.
- Interfaces with the body—Ensuring the design of the implanted device interfaces with the human body in a manner that provides the best function and performance without causing additional harm to the patient.
- Market hurdles—Demonstrating the implanted medical device offers real diagnostic or therapeutic results with a reasonable cost compared to other treatments.
- Regulatory hurdles—Ensuring that design and process controls are sufficient to meet the demands of an U.S. Food and Drug Administration PMA or EU Technical Dossier process.
- Stigma—To some extent, implanted devices have a stigma because they’re implanted inside the body and they have to show effectiveness. You can’t do a “me-too” implanted device anymore, really, because you have to show effectiveness. Medical devices now obviously have a hurdle that is more significant than it was in the past. And the risk of a surgery is always high. I think the last thing a person wants to hear is we’re going to cut you open and put something inside of you. Certainly, minimally invasive surgery helps with that but keep in mind that other aspects of medicine are improving, too. But our customers still tend to think about implanted devices as a last resort-type of therapy.
Highley: Typically it’s the regulatory approval process. I believe these devices are relatively straightforward—we know them very well, they’re characterized by the design and the construction. It’s just that the regulatory approval process is pretty extensive and quite expensive. There’s a significant amount of testing and clinical data that has to be collected to get approval for a product. Many companies will go to Europe to launch their product first. This was a trend discussed when I was over on the other side of the business—the traditional Class II device business. But many of these Class III devices start off in Europe because the regulatory process is a bit easier and actually helps them work toward FDA approval, which can take years and a significant financial investment. It is not uncommon to spend over one hundred million dollars to get a product developed and launched in the United States. It’s an expensive endeavor.
Barbella: How have you addressed these challenges?
Cameron, Gregory, Lambert: Our team stays current with evolving regulations globally to support our materials in our customers’ markets. NuSil has more than 20 years’ of experience providing MAF and DMF supported long-term implantable materials to provide our customers’ path for approval.
Fine: Our company has a well seasoned medical device engineering staff that has been working on implanted medical devices for many years. They work within an environment that has a robust quality system to ensure that the activities meet regulatory and quality standards throughout the world. We work with companies, universities, and medical institutions around the world applying our engineering skills, and gaining exposure and additional expertise with a wide variety of implanted and external medical device technologies.
Barbella: How are implantable device designers and/or manufacturers addressing the demand for miniaturization and ever-smaller products? Is there a limit to how small an implantable device can be?
Cameron, Gregory, Lambert: Who would have thought that pacemakers, which were initially the size of a hockey puck when first introduced, would ever be the size of a pill—but they have. Technology in microelectronics, semiconductors, and microfluidics will contribute to increased miniaturization. We don’t think we’ve reached the limit to how small. There are certainly constraints in physics.
Fine: At some point there is a limit to how small an implantable device can be by the nature of what it is trying to accomplish and how it needs to interface with the body to accomplish its function. For active implantable devices, the size may be limited by the amount of energy required for the device to function over several years. Battery technology and density has improved to allow for smaller batteries. Also, wireless energy transfer systems have allowed for smaller rechargeable batteries or devices that do not require an internal primary battery. In addition, chip level circuit integration and novel electronics packaging have allowed engineers to develop smaller active implantable devices. Other implantable devices may have particular strength or robustness requirements, such as with orthopedic implants. As materials improve, the mass of these devices can be reduced.
Highley: A lot of the innovation going on in this space today is miniaturization. Unfortunately, the market tends to move at a glacial pace because change is difficult to implement and there’s a lot of technology that has to be developed. The way to think about miniaturization, I believe, is to get one of these neurostimulators to a point where you can implant it minimally invasively. When that happens the market should expand rapidly. Take for example, a traditional-sized neurostimulator that’s been developed to treat diabetes. Most people would say, “I don’t want this large implant in me to treat my diabetes, I’m going to treat it with medication.” But if I can get the implant small enough to implant minimally invasively, most people will say, “Yes, now that is a viable option for me.” So ideally, we’re working toward these miniaturized, minimally invasively implantable devices that I believe will expand the market rapidly.
Miniaturization is not just in neuromodulation, it can be expanded to other markets as well, including ventricular assist devices. Of course, these aren’t going to get to the size that is minimally invasively implantable like a nerve stimulator, but Cirtec is developing fully implantable ventricular assist devices, meaning there’s no percutaneous line sticking out of your abdomen tying it to an external battery pack. The battery is fairly small and implanted internally. We believe when you’re able to miniaturize these devices, implant them less invasively and have them fully implanted, eliminating the external battery pack, that market will expand as well. So miniaturization is driving a lot of innovation in the active implantable space.
Of course there’s a limit to how small these devices need to be. You need it to be the size of something you can implant minimally invasively through a needle. Let’s use a grain of rice as an example. Is there any reason to go to a size half a grain of rice? Or a third of a grain of rice? Probably not. There’s a point where you may not need to get any smaller for these kinds of devices, but it doesn’t mean people will stop trying.
Barbella: The assimilation of electronics in implantable medical devices has raised hacking concerns within the industry. Is this a legitimate threat? Please explain your answer.
Cameron, Gregory, Lambert: There are implantable devices that function purely on physical/mechanical principles which would have no concern over hacking. For example, hydrocephalic shunts manage function purely on hydraulic pressure.
Fine: Anytime a device, medical or otherwise, can communicate with a network of any type, hacking is a legitimate threat. As more medical devices interact with the internet for data capture or remote programming, robust security measures need to be considered.
Highley: Yes, I think it’s absolutely a legitimate threat but I believe there are, and will be, smart people and companies out there that can add technology to the design to make these devices safe and secure from hacking and other malicious manipulation. Cirtec is working with a few companies that are spearheading security in these implantable devices. This is another great example of the convergence of non-traditional business partners now innovating more and more in the medical device industry; smartphones, apps, wireless communication, security, and privacy will all play a critical role in the future. We can’t move quite as quickly as the consumer world, so in order to include security into a device you have to design it early, develop it, validate it, and obtain all your [regulatory] approvals with it already integrated. Cirtec is working on this now because we know companies will demand it in the future. It is a legitimate threat but it’s slow to implement in this environment.
Barbella: How can implantable medical device developers safeguard their product(s) from unauthorized access, or hackers?
Fine: There are several strategies that can be employed to safeguard medical products from hacking. Medical device developers can leverage existing methodologies such as encryption or multiple level authentication to restrict access to wireless communication. Another strategy may be to implement a totally proprietary communication scheme that does not depend on known standards.
Barbella: Is it reasonable to expect that most—if not all—external medical technologies will eventually become implantable? Why or why not?
Cameron, Gregory, Lambert: There are many external medical devices that are needed for only a short period of time. On the other hand, there are many organs that are transplanted today that we could imagine being replaced with implantable devices.
Fine: I believe that many external technologies will always stay external. Implantable devices are best suited to long-term chronic conditions that cannot be treated easily by other means. Drugs or other means may be a more cost-effective alternative, or present less patient risk than an implanted medical device.
In healthcare, we attempt to either diagnose or treat a particular condition or disease. As some of the non-implantable diagnostic technology improves, it negates the need for an implant. A cardiac loop detector is a good example. There’s a long-term skin patch from LifeWatch that detects arrhythmias, and is replaceable after a few weeks or months. Personally, I’d rather have something on my skin than in my body. Companies also are working on both internal and external versions of glucose monitors.
I think there’s always going to be the yin and yang of technology, and the two are going to fight each other. But at the end of the day, from a use preference point of view, an external device is going to be preferable to an implantable one. Even so, there are some applications where being inside the body is advantageous because the condition or disease can be treated more effectively near the source. As much as implants advance, external devices are going to advance also. At the end of the day, it’s probably more desirable not to cut open the body and put something inside of it.
Barbella: Is it a prerequisite now that implantable medical devices have the ability to communicate wirelessly with consumer electronics like smartphones or tablets? Why or why not?
Cameron, Gregory, Lambert: Having a device that communicates to an external phone or tablet has proven to be useful in some therapies. In addition, it may be useful to detect that a device is present, as well as more information on the device or patient through RFID technology.
Fine: Because there are so many disease states that can be treated by implantable devices, I don’t believe that as a blanket statement medical devices need to communicate with consumer wireless devices. It is likely that many orthopedic implants will remain without a means to communicate outside the body. However, given the advancement of IoT technology, many patients and clinicians involved with implanted medical devices would derive a benefit from such connectivity. For example, implanted diagnostic devices, such as cardiac loop recorders or long term glucose monitors, would benefit from connectivity available to the patient and clinician. Other implanted devices with which a patient or clinician may benefit from interfacing regularly include products such as insulin pumps, pain treatment devices, and a number of neurological treatment devices.
This issue is similar to internal-external (device) argument, whereas one of the best ways to keep something secure is to not connect it to another source. There’s going to be multiple camps. I think what’s going to happen is there will always a camp that believes you have to have the latest bells and whistles, regardless of whether you use them. A lot of the features on early pacemakers were there because the competition had it and not because anybody actually used it. We do a lot of work in implantable drug infusion pumps. Even though constant flow pumps kind of failed and Medtronic pushed programmability, 90 percent of these programmable pumps were set for one rate and left alone. Maybe some of that is you have to have the feature because somebody else has the feature. But it really should be a function of whether or not there is actual value by having the information from the device be available to somebody externally. If that’s the case, then a tablet or going through the cloud is often a good way to do it.
Highley: I believe that’s where these devices are heading, and many Class II devices have been there for years. We see more and more requests for devices that can work with and communicate with other external devices. The problem here is the lifecycle of the external device. For instance, let’s say the obsolescence on an iPhone is 18 months. It’s very tough to design an implantable device that works with an iPhone 6. Two iterations later it’s no longer relevant; you’ll have to continuously update and test your software so that it works with the next-generation phone. I think people have to find a way around doing that all the time. It’s a challenge. People are looking to ensure their devices can communicate with tablets and phones securely but it’s a huge challenge because of the lifecycle of the devices that they’re connecting to, and controlling a potentially lifesaving device is a bit different than updating Candy Crush. The companies that can solve these problems, and frankly, profit from them, are not traditionally aligned to the medical device industry’s regulatory environment. They’re focused on their consumer markets, and it is a steep learning curve to understand the requirements for medical devices and FDA scrutiny. Ultimately, the regulatory bodies, medical device industry leaders, and Silicon Valley experts need to collaborate on this issue. It has started in small pockets of the industry but it will take some time for it to become standardized.