06.02.15
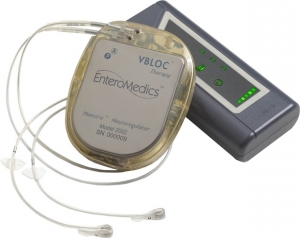
The first commercial implant of EnteroMedics’ Maestro rechargeable system has been completed in the United States. The system, which delivers the St. Paul, Minn.-based company’s proprietary Vbloc neurometabolic therapy, uses neuroblocking to treat obesity.
The procedure was performed at Tufts Medical Center in Boston, Mass., by surgeon Sajani Shah, M.D. The surgery reportedly proceeded without incident and the patient returned home on the same day as the surgery, is recovering well and has returned to work full-time.
“Until recently, there was a great unmet need for new, alternative treatment options for obese patients for whom behavior modification, pharmaceutical options or anatomy altering/restricting surgical procedures either did not work, or were not viable options,” said Shah.
“Earlier this year, the [U.S. Food and Drug Administration] approved a first-of-its-kind treatment that offers a neuroscience-based approach to the treatment of obesity, differentiating it from traditional weight loss surgical options. As a member of the team that brought this new device through the clinical trial phase, I am pleased to have performed the first U.S. commercial implant of Vbloc Therapy. At Tufts Medical Center, we are committed to staying on the leading edge of new technologies that may benefit our patients.”
Vbloc therapy intermittently blocks intra-abdominal vagus nerve signaling, or transmission of messages involving food intake and processing, between the brain and stomach using the Maestro rechargeable system, which works somewhat like a pacemaker. In January of this year, the system was approved for adults within a certain body mass index range with a related health condition such as high blood pressure or high cholesterol levels, and who have tried to lose weight in a supervised weight management program within the past five years.
“The first commercial implant of Vbloc therapy marks a significant milestone for EnteroMedics and the millions of individuals with obesity who may be seeking different ways of achieving safe, effective weight loss,” Mark B. Knudson, Ph.D., president and CEO of EnteroMedics, said in prepared remarks. “We are pleased with the response to Vbloc therapy from patients and physicians alike and remain focused on the continued development of partnerships with bariatric centers of excellence and on driving reimbursement for Vbloc therapy to ensure its widespread availability in the United States.”
The procedure was performed at Tufts Medical Center in Boston, Mass., by surgeon Sajani Shah, M.D. The surgery reportedly proceeded without incident and the patient returned home on the same day as the surgery, is recovering well and has returned to work full-time.
“Until recently, there was a great unmet need for new, alternative treatment options for obese patients for whom behavior modification, pharmaceutical options or anatomy altering/restricting surgical procedures either did not work, or were not viable options,” said Shah.
“Earlier this year, the [U.S. Food and Drug Administration] approved a first-of-its-kind treatment that offers a neuroscience-based approach to the treatment of obesity, differentiating it from traditional weight loss surgical options. As a member of the team that brought this new device through the clinical trial phase, I am pleased to have performed the first U.S. commercial implant of Vbloc Therapy. At Tufts Medical Center, we are committed to staying on the leading edge of new technologies that may benefit our patients.”
Vbloc therapy intermittently blocks intra-abdominal vagus nerve signaling, or transmission of messages involving food intake and processing, between the brain and stomach using the Maestro rechargeable system, which works somewhat like a pacemaker. In January of this year, the system was approved for adults within a certain body mass index range with a related health condition such as high blood pressure or high cholesterol levels, and who have tried to lose weight in a supervised weight management program within the past five years.
“The first commercial implant of Vbloc therapy marks a significant milestone for EnteroMedics and the millions of individuals with obesity who may be seeking different ways of achieving safe, effective weight loss,” Mark B. Knudson, Ph.D., president and CEO of EnteroMedics, said in prepared remarks. “We are pleased with the response to Vbloc therapy from patients and physicians alike and remain focused on the continued development of partnerships with bariatric centers of excellence and on driving reimbursement for Vbloc therapy to ensure its widespread availability in the United States.”