PR Newswire09.30.20
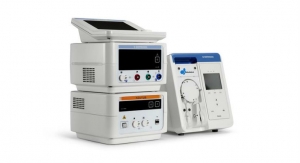
SentiAR, a pioneer in visualization technologies for surgical applications announces U.S. Food and Drug Administration (FDA) 510(k) clearance of its groundbreaking CommandEP system, the first holographic guidance system to be used during an invasive cardiac procedure. The CommandEP system allows electrophysiologists to visualize 3D electroanatomic models in real-time, via a wearable headset and offer a truly handsfree control interface providing superior visualization, allowing them to navigate accurately within the heart during a cardiac ablation procedure.
The CommandEP system receives imaging data from the electroanatomic mapping systems and creates a 3D holographic image via SentiAR's proprietary data flow and visualization algorithms. This patented system presents a 3D image of the patient's heart showing the real-time position of the catheters within to the physicians via a wearable headset, currently Microsoft HoloLens. Additionally, SentiAR deploys its unique, trade-secret AR engine to deliver uncompromising performance.
SentiAR successfully completed a clinical study with the CommandEP system at St. Louis Children's and Barnes Jewish Hospitals. During the procedure with CommandEP, physicians were asked to perform a navigation study by targeting specific points within the patient anatomy. Each navigation exercise was completed with the CommandEP system and repeated again with standard visualization tools. The results of the comparative analysis showed a 44 percent reduction in mean navigation error with the CommandEP system over standard visualization tools. The results of this study were published in Journal of American College of Cardiology: Clinical Electrophysiology.
"The addition of SentiAR's holographic guidance system provides electrophysiologists with a game-changing platform for mapping and interventional procedures," said John K. Triedman, M.D., Chief of Cardiac Electrophysiology at Boston Children's Hospital. "The ability to see the models in 3D in real-time, to look inside chambers and around corners while moving and rotating the model allows us to gain a better and more precise understanding of cardiac anatomy. We anticipate being able to easily integrate the CommandEP system with our existing EP lab workflows. The powerful tools CommandEP provides have real potential to allow electrophysiologists to perform more accurate procedures."
"CommandEP represents a huge leap forward in cardiac ablation technology," said Paul Buckman, member of the board of directors of SentiAR. "Our clinical and strategic partners are thrilled with the patient data showing improved navigation, and together we look forward to putting CommandEP into the hands of electrophysiologists worldwide."
"SentiAR's holographic ablation guidance system is our first of many products that will revolutionize interventional procedures," said Berk Tas, CEO of SentiAR. "SentiAR provides physicians superhuman capabilities to integrate their digital environments so they can focus on patients and their treatment."
The CommandEP system is now available for sale in the United States with distribution expected to begin in 2021.
BioGenerator, the investment arm of BioSTL, and Cultivation Capital Life Sciences Fund are lead investors in SentiAR.
The CommandEP system receives imaging data from the electroanatomic mapping systems and creates a 3D holographic image via SentiAR's proprietary data flow and visualization algorithms. This patented system presents a 3D image of the patient's heart showing the real-time position of the catheters within to the physicians via a wearable headset, currently Microsoft HoloLens. Additionally, SentiAR deploys its unique, trade-secret AR engine to deliver uncompromising performance.
SentiAR successfully completed a clinical study with the CommandEP system at St. Louis Children's and Barnes Jewish Hospitals. During the procedure with CommandEP, physicians were asked to perform a navigation study by targeting specific points within the patient anatomy. Each navigation exercise was completed with the CommandEP system and repeated again with standard visualization tools. The results of the comparative analysis showed a 44 percent reduction in mean navigation error with the CommandEP system over standard visualization tools. The results of this study were published in Journal of American College of Cardiology: Clinical Electrophysiology.
"The addition of SentiAR's holographic guidance system provides electrophysiologists with a game-changing platform for mapping and interventional procedures," said John K. Triedman, M.D., Chief of Cardiac Electrophysiology at Boston Children's Hospital. "The ability to see the models in 3D in real-time, to look inside chambers and around corners while moving and rotating the model allows us to gain a better and more precise understanding of cardiac anatomy. We anticipate being able to easily integrate the CommandEP system with our existing EP lab workflows. The powerful tools CommandEP provides have real potential to allow electrophysiologists to perform more accurate procedures."
"CommandEP represents a huge leap forward in cardiac ablation technology," said Paul Buckman, member of the board of directors of SentiAR. "Our clinical and strategic partners are thrilled with the patient data showing improved navigation, and together we look forward to putting CommandEP into the hands of electrophysiologists worldwide."
"SentiAR's holographic ablation guidance system is our first of many products that will revolutionize interventional procedures," said Berk Tas, CEO of SentiAR. "SentiAR provides physicians superhuman capabilities to integrate their digital environments so they can focus on patients and their treatment."
The CommandEP system is now available for sale in the United States with distribution expected to begin in 2021.
BioGenerator, the investment arm of BioSTL, and Cultivation Capital Life Sciences Fund are lead investors in SentiAR.