GlobeNewswire07.22.20
Integra LifeSciences Holdings Corporation has received FDA clearance for a specific indication for neurosurgery for its CUSA Clarity Ultrasonic Surgical Aspirator System. The announcement occurs on Glioblastoma Awareness Day, a day dedicated to elevating public understanding of this aggressive and difficult to treat form of cancer.
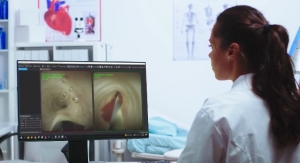
While the CUSA system has been used for over 40 years* in neurosurgical applications, this specific neurosurgery indication shows CUSA Clarity can be used safely and effectively in neurosurgery for resection of tumors ranging from soft to firm consistencies, which includes removal of primary and secondary malignant, and benign brain and spinal tumors, including but not limited to, meningiomas and gliomas. FDA clearance is based on a wealth of peer-reviewed clinical publications and 40 years of surgical cases involving resection of brain and spinal tumors.
“Integra has a long history of developing technologies that meet surgeon’s needs for enhanced surgical performance, especially for longer and tougher cases, such as brain tumor resections,” said Mike McBreen, executive vice president and president, Codman Specialty Surgical, Integra LifeSciences. “This specific indication represents our continued commitment to offering neurosurgeons safe and effective products that help them achieve the best outcomes for their patients.”
More than 700,000 patients in the United States are living with some form of brain tumor. The most prevalent types of brain tumor in adults are meningiomas and gliomas (such as glioblastoma, ependymoma, astrocytoma, and oligodendroglioma), which make up 81 percent of malignant brain tumors in adults.1
Tumor resection surgery is a common approach to treating cancer patients and surgical resection of a tumor can be performed for many types of cancer. The goal of surgery is to remove the entire tumor, or as much of the tumor as possible.2 CUSA Clarity Ultrasonic Surgical Aspirator System is an ultrasonically vibrating surgical device which, in combination with irrigation and aspiration, fragments, emulsifies and removes unwanted tissue. It allows for selective dissection of target tissue like tumors while preserving vessels, ducts and other delicate structures.
References
1 American Brain Tumor Association.
2 Temple Health Tumor Resection Surgery.
* Publications submitted to FDA for CUSA neuro use dated back to 1978
While the CUSA system has been used for over 40 years* in neurosurgical applications, this specific neurosurgery indication shows CUSA Clarity can be used safely and effectively in neurosurgery for resection of tumors ranging from soft to firm consistencies, which includes removal of primary and secondary malignant, and benign brain and spinal tumors, including but not limited to, meningiomas and gliomas. FDA clearance is based on a wealth of peer-reviewed clinical publications and 40 years of surgical cases involving resection of brain and spinal tumors.
“Integra has a long history of developing technologies that meet surgeon’s needs for enhanced surgical performance, especially for longer and tougher cases, such as brain tumor resections,” said Mike McBreen, executive vice president and president, Codman Specialty Surgical, Integra LifeSciences. “This specific indication represents our continued commitment to offering neurosurgeons safe and effective products that help them achieve the best outcomes for their patients.”
More than 700,000 patients in the United States are living with some form of brain tumor. The most prevalent types of brain tumor in adults are meningiomas and gliomas (such as glioblastoma, ependymoma, astrocytoma, and oligodendroglioma), which make up 81 percent of malignant brain tumors in adults.1
Tumor resection surgery is a common approach to treating cancer patients and surgical resection of a tumor can be performed for many types of cancer. The goal of surgery is to remove the entire tumor, or as much of the tumor as possible.2 CUSA Clarity Ultrasonic Surgical Aspirator System is an ultrasonically vibrating surgical device which, in combination with irrigation and aspiration, fragments, emulsifies and removes unwanted tissue. It allows for selective dissection of target tissue like tumors while preserving vessels, ducts and other delicate structures.
References
1 American Brain Tumor Association.
2 Temple Health Tumor Resection Surgery.
* Publications submitted to FDA for CUSA neuro use dated back to 1978