Business Wire06.07.18
iSchemaView, the leader in cerebrovascular imaging analysis, has received final clearance from the U.S. Food and Drug Administration (FDA) for RAPID CTA, the company’s 3D imaging solution for Computed Tomography Angiography. RAPID CTA is the newest addition to iSchemaView’s industry leading neuroimaging platform—including RAPID CTP and RAPID MRI—which is designed to provide physicians with fast, fully-automated, elegant and easy-to-interpret imaging that facilitates clinical decision making around cerebrovascular disease, such as stroke.
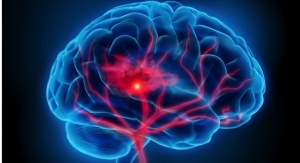
In simplest terms, CTA provides visualization of blood vessels in the brain. Using an injectable contrast agent, high-tech X-ray imaging and sophisticated computer analysis, a computed tomography (CT) angiogram provides physicians with detailed and accurate images of the vasculature. However, interpreting standard CTA output is a cumbersome time-consuming process.
RAPID CTA automatically provides clear, easy to interpret CTA maps which include a colored overlay to identify brain regions with reduced blood vessel density. The severity of reduction can be readily visualized by a simple, four-color-coded scale. Additionally, a 3D reconstruction of the vasculature allows physicians to rotate the image for optimal viewing of the vessels from multiple angles. All of this in a few minutes. RAPID CTA is a phenomenal tool for all doctors involved in neurovascular emergencies—from the ER doctor making decisions about patient transfer to the neurointerventionalist getting ready for an incoming thrombectomy procedure.
“Our goal is to provide healthcare professionals around the world with the most comprehensive and intuitive suite of imaging solutions possible,” said Don Listwin, CEO of iSchemaView. “With new FDA clearance and the release of RAPID CTA, we are making good on that goal. We ensure physicians can take advantage of RAPID’s power whether they choose plain CT, CT Angiography, CT Perfusion or MR diffusion and perfusion. It’s all about offering them the right imaging solution choice, for the right patient at the right time.”
In an effort to save valuable diagnosis and treatment time, RAPID CTA also offers remote viewing for physicians in the hospital, at home or on the road. Image maps are immediately available to doctors for review on any desktop or mobile device.
RAPID CTA maps provide an intuitive and easy to interpret view of the brain vessels, helping physicians with:
“Many eligible stroke patients with large vessel occlusions must be quickly transferred to a comprehensive stroke center. The new automated RAPID CTA tool makes it easy for community hospitals to quickly identify potential candidates for treatment and for experts at the comprehensive centers to verify that the patient is appropriate for transfer,” said Greg Albers, professor of Neurology at Stanford University, director of the Stanford Stroke Center and cofounder of iSchemaView. “With the addition of CTA to RAPID’s FDA indications, hospitals and clinics of all sizes and locations can now use the platform to immediately review images from wherever they are. That’s powerful.”
iSchemaView is the leader in cerebrovascular imaging analysis, delivering software tools used to identify and treat stroke patients. iSchemaView’s RAPID is the most advanced brain imaging platform, and in clinical trials, under IRB, RAPID has been shown to aid in the selection of patients in early and late window stroke trials, including SWIFT PRIME, EXTEND IA, DAWN and DEFUSE 3. In addition to achieving the best clinical outcomes and largest treatment effects ever obtained, these studies led to new American Heart Association and American Stroke Association guidelines, and have altered the management of acute stroke around the world.
In simplest terms, CTA provides visualization of blood vessels in the brain. Using an injectable contrast agent, high-tech X-ray imaging and sophisticated computer analysis, a computed tomography (CT) angiogram provides physicians with detailed and accurate images of the vasculature. However, interpreting standard CTA output is a cumbersome time-consuming process.
RAPID CTA automatically provides clear, easy to interpret CTA maps which include a colored overlay to identify brain regions with reduced blood vessel density. The severity of reduction can be readily visualized by a simple, four-color-coded scale. Additionally, a 3D reconstruction of the vasculature allows physicians to rotate the image for optimal viewing of the vessels from multiple angles. All of this in a few minutes. RAPID CTA is a phenomenal tool for all doctors involved in neurovascular emergencies—from the ER doctor making decisions about patient transfer to the neurointerventionalist getting ready for an incoming thrombectomy procedure.
“Our goal is to provide healthcare professionals around the world with the most comprehensive and intuitive suite of imaging solutions possible,” said Don Listwin, CEO of iSchemaView. “With new FDA clearance and the release of RAPID CTA, we are making good on that goal. We ensure physicians can take advantage of RAPID’s power whether they choose plain CT, CT Angiography, CT Perfusion or MR diffusion and perfusion. It’s all about offering them the right imaging solution choice, for the right patient at the right time.”
In an effort to save valuable diagnosis and treatment time, RAPID CTA also offers remote viewing for physicians in the hospital, at home or on the road. Image maps are immediately available to doctors for review on any desktop or mobile device.
RAPID CTA maps provide an intuitive and easy to interpret view of the brain vessels, helping physicians with:
- Clinical decision making
- Patient triage
- Collaboration between community hospitals and specialists
- Appropriate patient transfers to specialty centers
“Many eligible stroke patients with large vessel occlusions must be quickly transferred to a comprehensive stroke center. The new automated RAPID CTA tool makes it easy for community hospitals to quickly identify potential candidates for treatment and for experts at the comprehensive centers to verify that the patient is appropriate for transfer,” said Greg Albers, professor of Neurology at Stanford University, director of the Stanford Stroke Center and cofounder of iSchemaView. “With the addition of CTA to RAPID’s FDA indications, hospitals and clinics of all sizes and locations can now use the platform to immediately review images from wherever they are. That’s powerful.”
iSchemaView is the leader in cerebrovascular imaging analysis, delivering software tools used to identify and treat stroke patients. iSchemaView’s RAPID is the most advanced brain imaging platform, and in clinical trials, under IRB, RAPID has been shown to aid in the selection of patients in early and late window stroke trials, including SWIFT PRIME, EXTEND IA, DAWN and DEFUSE 3. In addition to achieving the best clinical outcomes and largest treatment effects ever obtained, these studies led to new American Heart Association and American Stroke Association guidelines, and have altered the management of acute stroke around the world.