Globe Newswire04.10.18
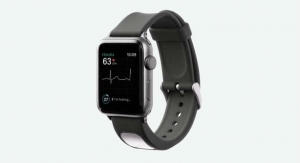
iRhythm Technologies Inc., a digital healthcare solutions company focused on the advancement of cardiac care, has announced primary results of the mHealth Screening to Prevent Strokes (mSToPS) study that evaluated detection of silent atrial fibrillation (AF) in high-risk individuals using Zio by iRhythm.
Electrocardiogram (ECG) recording and analysis were carried out using the U.S. Food and Drug Administration-cleared Zio by iRhythm ambulatory monitoring patch. After four months, the new incidence of AF was 5.1 percent among patients who were immediately monitored with the Zio Service compared with 0.6 percent in patients who were assigned to a delayed monitoring group and had not yet worn the Zio monitor. At one year, primary results showed that AF was newly diagnosed in 6.3 percent of patients who were actively monitored by the Zio service versus 2.3 percent in the observational control group receiving routine care. In addition, 4 percent of patients in the Zio monitored group were found to have potentially actionable arrhythmias other than AF including ventricular tachycardia, pause, AV block, and symptomatic supraventricular tachycardia.
Monitoring with Zio led to significant change in clinical treatment of the actively monitored group including initiation of anticoagulation therapies (5.4 percent), antiarrhythmic medications (0.8 percent), and pacemaker placement (0.7 percent). Patients receiving anticoagulation for AF appeared to fulfill current clinical guidelines for appropriateness and need. Mean wear time for the Zio patch was 12 days with 98 percent analyzable ECG data.
Researchers at the Scripps Translational Science Institute (STSI) conducted the study in partnership with collaborators, Aetna and Janssen Pharmaceuticals. The study design demonstrated the digital solution enabled by Zio effectively monitored a large and geographically disperse population of patients who had risk factors for AF. “By fully deploying the digital infrastructure to reimagine how clinical trials can be performed, we hope that our efforts will be a useful template for highly efficient remote enrollment and engagement with participants,” said Eric Topol, M.D., founder and director of STSI at the Scripps Research Institute.
The study involved 5,214 eligible Aetna members who were identified through claims data to have risk factors for AF but had not been previously diagnosed. Of the total study participants, 1,738 individuals were enrolled via a web-based platform to undergo either immediate or delayed active ECG monitoring at home for up to four weeks with a Zio XT patch monitor (two-week monitoring periods spaced four months apart). Each monitored participant was matched with two non-monitored participants with a similar CHA2DS2-VASc, a standardized stroke-risk assessment score, to act as controls. The study looked at time to first diagnosis of AF and its clinical consequences for the active monitoring cohort as well as the cohort undergoing usual care.
“Trials like mSToPS play a critical role in defining the population of at-risk patients where targeted detection with extended continuous ambulatory monitoring is beneficial,” said Judy Lenane, R.N., MHA, executive vice president of operations and chief clinical officer of iRhythm. “This study further establishes Zio by iRhythm as a patient-friendly, efficient tool for detecting asymptomatic or silent atrial fibrillation in high risk patients, leading to diagnosis in a single test and accelerated care.”
Utilizing the digital Zio monitoring service by iRhythm enabled the team to approach a large, geographically diverse population of at-risk individuals and include people who otherwise may have no access to participation in clinical trials because they do not live close to a research center.
“The future of medicine, especially preventive care, will be driven by digital sensors such as Zio by iRhythm,” said Steven Steinhubl, M.D., director of digital medicine at STSI and principal investigator of the mSToPS trial. “Our results show that the Zio extended continuous cardiac monitoring system enabled us to detect AF in asymptomatic patients.”
Atrial fibrillation is a quivering or irregular heartbeat, also known as an arrhythmia, which can lead to blood clots, stroke, heart failure and other heart-related complications. Normally, the heart contracts and relaxes to a regular beat. In AF, the upper chambers of the heart (the atria) beat irregularly instead of beating effectively to move blood into the ventricle.
AF is associated with a five-fold increase in the risk of stroke, with these strokes tending to be more severe and are associated with higher mortality.1 However, approximately one-third of those who have AF are not aware that they have it.2 Asymptomatic AF is referred to as being “silent” and there are certain risk factors like high blood pressure, diabetes and asthma that increase an individual’s likelihood for developing it.
iRhythm is redefining the way cardiac arrhythmias are clinically diagnosed. The company combines wearable biosensor devices worn for up to 14 days and cloud-based data analytics with powerful proprietary algorithms that distill data from millions of heartbeats into clinically actionable information. The company believes improvements in arrhythmia detection and characterization have the potential to change clinical management of patients.
References
1. Circulation. 2017;135:00–00. DOI: 10.1161/CIR.0000000000000485. Heart Disease and Stroke Statistics—2017 Update.
2. Friberg L, Rosenqvist M, Lindgren A, Terént A, Norrving B, Asplund K. High prevalence of atrial fibrillation among patients with ischemic stroke. Stroke 2014;45:2599-605.
Electrocardiogram (ECG) recording and analysis were carried out using the U.S. Food and Drug Administration-cleared Zio by iRhythm ambulatory monitoring patch. After four months, the new incidence of AF was 5.1 percent among patients who were immediately monitored with the Zio Service compared with 0.6 percent in patients who were assigned to a delayed monitoring group and had not yet worn the Zio monitor. At one year, primary results showed that AF was newly diagnosed in 6.3 percent of patients who were actively monitored by the Zio service versus 2.3 percent in the observational control group receiving routine care. In addition, 4 percent of patients in the Zio monitored group were found to have potentially actionable arrhythmias other than AF including ventricular tachycardia, pause, AV block, and symptomatic supraventricular tachycardia.
Monitoring with Zio led to significant change in clinical treatment of the actively monitored group including initiation of anticoagulation therapies (5.4 percent), antiarrhythmic medications (0.8 percent), and pacemaker placement (0.7 percent). Patients receiving anticoagulation for AF appeared to fulfill current clinical guidelines for appropriateness and need. Mean wear time for the Zio patch was 12 days with 98 percent analyzable ECG data.
Researchers at the Scripps Translational Science Institute (STSI) conducted the study in partnership with collaborators, Aetna and Janssen Pharmaceuticals. The study design demonstrated the digital solution enabled by Zio effectively monitored a large and geographically disperse population of patients who had risk factors for AF. “By fully deploying the digital infrastructure to reimagine how clinical trials can be performed, we hope that our efforts will be a useful template for highly efficient remote enrollment and engagement with participants,” said Eric Topol, M.D., founder and director of STSI at the Scripps Research Institute.
The study involved 5,214 eligible Aetna members who were identified through claims data to have risk factors for AF but had not been previously diagnosed. Of the total study participants, 1,738 individuals were enrolled via a web-based platform to undergo either immediate or delayed active ECG monitoring at home for up to four weeks with a Zio XT patch monitor (two-week monitoring periods spaced four months apart). Each monitored participant was matched with two non-monitored participants with a similar CHA2DS2-VASc, a standardized stroke-risk assessment score, to act as controls. The study looked at time to first diagnosis of AF and its clinical consequences for the active monitoring cohort as well as the cohort undergoing usual care.
“Trials like mSToPS play a critical role in defining the population of at-risk patients where targeted detection with extended continuous ambulatory monitoring is beneficial,” said Judy Lenane, R.N., MHA, executive vice president of operations and chief clinical officer of iRhythm. “This study further establishes Zio by iRhythm as a patient-friendly, efficient tool for detecting asymptomatic or silent atrial fibrillation in high risk patients, leading to diagnosis in a single test and accelerated care.”
Utilizing the digital Zio monitoring service by iRhythm enabled the team to approach a large, geographically diverse population of at-risk individuals and include people who otherwise may have no access to participation in clinical trials because they do not live close to a research center.
“The future of medicine, especially preventive care, will be driven by digital sensors such as Zio by iRhythm,” said Steven Steinhubl, M.D., director of digital medicine at STSI and principal investigator of the mSToPS trial. “Our results show that the Zio extended continuous cardiac monitoring system enabled us to detect AF in asymptomatic patients.”
Atrial fibrillation is a quivering or irregular heartbeat, also known as an arrhythmia, which can lead to blood clots, stroke, heart failure and other heart-related complications. Normally, the heart contracts and relaxes to a regular beat. In AF, the upper chambers of the heart (the atria) beat irregularly instead of beating effectively to move blood into the ventricle.
AF is associated with a five-fold increase in the risk of stroke, with these strokes tending to be more severe and are associated with higher mortality.1 However, approximately one-third of those who have AF are not aware that they have it.2 Asymptomatic AF is referred to as being “silent” and there are certain risk factors like high blood pressure, diabetes and asthma that increase an individual’s likelihood for developing it.
iRhythm is redefining the way cardiac arrhythmias are clinically diagnosed. The company combines wearable biosensor devices worn for up to 14 days and cloud-based data analytics with powerful proprietary algorithms that distill data from millions of heartbeats into clinically actionable information. The company believes improvements in arrhythmia detection and characterization have the potential to change clinical management of patients.
References
1. Circulation. 2017;135:00–00. DOI: 10.1161/CIR.0000000000000485. Heart Disease and Stroke Statistics—2017 Update.
2. Friberg L, Rosenqvist M, Lindgren A, Terént A, Norrving B, Asplund K. High prevalence of atrial fibrillation among patients with ischemic stroke. Stroke 2014;45:2599-605.