Business Wire03.14.18
AtriCure Inc., a developer of surgical treatments for atrial fibrillation (Afib) and left atrial appendage management, has named Scott Drake as board chairman, effective at the annual meeting in May of this year. Concurrent with this appointment, Richard Johnston, the company’s current chairman, and Elizabeth D. Krell, Ph.D., are planning to retire from the Board of Directors and are not standing for re-election.
“Scott’s proven executive experience managing growing businesses, achieving sustainable revenue growth and driving operational improvements have been invaluable to our organization over the past four years. I am delighted that AtriCure can further benefit from his leadership in his new role as board chairman” stated Johnson.
Drake has served as one of the company’s directors since September 2013. He was most recently the president and CEO of Spectranetics Corporation. Spectranetics, acquired by Philips in August of 2017, develops, manufactures and distributes single-use medical devices used in minimally invasive procedures within the cardiovascular system. Prior to joining Spectranetics, Drake served as senior vice president, Operations, of DaVita Inc., a U.S. provider of kidney care and dialysis, from 2009 to August 2011. Previously, Drake held several positions of increasing responsibility within various healthcare business units at Covidien plc over a period of 17 years, including: global business unit president, Respiratory and Monitoring Solutions; president, Valleylab (re-named as the Surgical Solutions Group); and vice president and general manager, Critical Care. Drake is an EY 2014 Entrepreneur of The Year. He also serves on the Board of Directors for Just Right Surgical and the Medical Device Manufacturers Association (MDMA). Drake has a bachelor of science degree in business administration from Miami University of Ohio.
“I would like to acknowledge Dick and Liz for their many contributions to our organization during their respective tenures on our Board,” stated Mike Carrel, president and CEO of AtriCure. “Dick’s direction as our longstanding chairman has been critical in navigating the company to our leading position in the market today. From the early days of the company’s inception, through a successful public offering, to bringing me on board as CEO, to managing the board composition, and to helping reshape the company over the past five plus years, Dick’s impact will be felt for many years to come. Liz has been instrumental in our regulatory and clinical strategies over the past ten years, bringing expertise in risk assessment and healthcare compliance. On behalf of our entire board, I thank them both for their truly valuable service and guidance.”
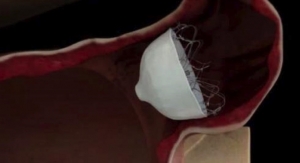
AtriCure Inc. provides technologies for the treatment of Afib and related conditions. Afib affects more than 33 million people worldwide. Electrophysiologists and cardiothoracic surgeons around the globe use AtriCure technologies for the treatment of Afib and reduction of Afib related complications. AtriCure’s Isolator Synergy Ablation System is the first and only medical device to receive U.S. Food and Drug Administration approval for the treatment of persistent Afib. AtriCure’s AtriClip Left Atrial Appendage Exclusion System products are the most widely sold LAA management devices worldwide, with more than 125,000 implanted to date.
“Scott’s proven executive experience managing growing businesses, achieving sustainable revenue growth and driving operational improvements have been invaluable to our organization over the past four years. I am delighted that AtriCure can further benefit from his leadership in his new role as board chairman” stated Johnson.
Drake has served as one of the company’s directors since September 2013. He was most recently the president and CEO of Spectranetics Corporation. Spectranetics, acquired by Philips in August of 2017, develops, manufactures and distributes single-use medical devices used in minimally invasive procedures within the cardiovascular system. Prior to joining Spectranetics, Drake served as senior vice president, Operations, of DaVita Inc., a U.S. provider of kidney care and dialysis, from 2009 to August 2011. Previously, Drake held several positions of increasing responsibility within various healthcare business units at Covidien plc over a period of 17 years, including: global business unit president, Respiratory and Monitoring Solutions; president, Valleylab (re-named as the Surgical Solutions Group); and vice president and general manager, Critical Care. Drake is an EY 2014 Entrepreneur of The Year. He also serves on the Board of Directors for Just Right Surgical and the Medical Device Manufacturers Association (MDMA). Drake has a bachelor of science degree in business administration from Miami University of Ohio.
“I would like to acknowledge Dick and Liz for their many contributions to our organization during their respective tenures on our Board,” stated Mike Carrel, president and CEO of AtriCure. “Dick’s direction as our longstanding chairman has been critical in navigating the company to our leading position in the market today. From the early days of the company’s inception, through a successful public offering, to bringing me on board as CEO, to managing the board composition, and to helping reshape the company over the past five plus years, Dick’s impact will be felt for many years to come. Liz has been instrumental in our regulatory and clinical strategies over the past ten years, bringing expertise in risk assessment and healthcare compliance. On behalf of our entire board, I thank them both for their truly valuable service and guidance.”
AtriCure Inc. provides technologies for the treatment of Afib and related conditions. Afib affects more than 33 million people worldwide. Electrophysiologists and cardiothoracic surgeons around the globe use AtriCure technologies for the treatment of Afib and reduction of Afib related complications. AtriCure’s Isolator Synergy Ablation System is the first and only medical device to receive U.S. Food and Drug Administration approval for the treatment of persistent Afib. AtriCure’s AtriClip Left Atrial Appendage Exclusion System products are the most widely sold LAA management devices worldwide, with more than 125,000 implanted to date.