GE Healthcare05.09.17
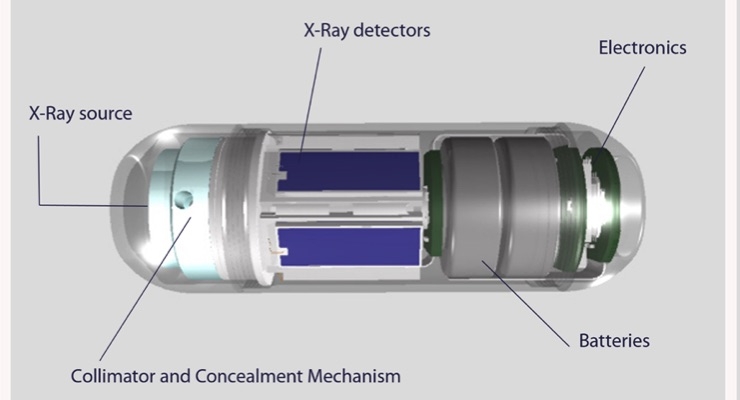
Check-Cap Ltd., a clinical-stage company engaged in the development of an ingestible capsule for preparation-free colorectal cancer screening, and GE Healthcare today announced the companies have successfully achieved the initial milestone in their ongoing collaboration to develop high-volume, X-ray capsule manufacturing capabilities. Specifically, the partnership involves X-ray sources produced at GE Healthcare using a customized manufacturing method passed all tests required to ensure compliance with C-Scan system specifications.
"We are very excited to have achieved success in the first critical phase of our collaboration with GE Healthcare, where we demonstrated the capability of our process to manufacture X-ray sources for the C-Scan system," said Bill Densel, CEO of Check-Cap. "We look forward to moving ahead with the development and validation of scalable production and integration processes that can provide capsules for future U.S. clinical trials and commercialization."
Colorectal cancer is the second leading cause of cancer death in the United States, with an estimated 135,000 diagnoses and 50,000 deaths in 2017.1 Despite evidence that screening can detect colorectal cancer and precancerous polyps, nearly one-third of the recommended adult population has never been screened. The C-Scan system is designed to improve the patient experience with screening by eliminating many unattractive requirements, such as bowel preparation, fasting, and sedation.
Using ultra-low dose X-ray and wireless communication technologies, the capsule generates information on the contours of the inside of the colon as it passes naturally. This information is used to create a 3D map of the colon, which allows physicians to look for polyps and other abnormalities. Designed to improve the patient experience and increase the willingness of individuals to participate in recommended colorectal cancer screening, C-Scan removes many frequently-cited barriers, such as laxative bowel preparation, invasiveness and sedation. The C-Scan system is currently not cleared for marketing in any jurisdiction.
"The results of this critical phase of the collaboration are incredibly exciting for patients and the future prevention of colorectal cancer," said Emmanuel Ligner, general manager of Core Imaging for GE Healthcare Life Sciences. "GE Healthcare will continue its collaboration with Check-Cap using our expertise in radiopharmaceutical and device manufacturing to bring the C-Scan system to clinicians and patients."
Check-Cap is a clinical-stage medical diagnostics company developing C-Scan, the first capsule-based system for preparation-free colorectal cancer screening.
GE Healthcare provides transformational medical technologies and services to meet the demand for increased access, enhanced quality and more affordable healthcare around the world. From medical imaging, software & IT, patient monitoring and diagnostics to drug discovery, biopharmaceutical manufacturing technologies and performance improvement solutions, GE Healthcare helps medical professionals deliver healthcare to their patients.
Reference:
1. "Key Statistics for Colorectal Cancer," American Cancer Society, https://www.cancer.org/cancer/colon-rectal-cancer/about/key-statistics.html
"We are very excited to have achieved success in the first critical phase of our collaboration with GE Healthcare, where we demonstrated the capability of our process to manufacture X-ray sources for the C-Scan system," said Bill Densel, CEO of Check-Cap. "We look forward to moving ahead with the development and validation of scalable production and integration processes that can provide capsules for future U.S. clinical trials and commercialization."
Colorectal cancer is the second leading cause of cancer death in the United States, with an estimated 135,000 diagnoses and 50,000 deaths in 2017.1 Despite evidence that screening can detect colorectal cancer and precancerous polyps, nearly one-third of the recommended adult population has never been screened. The C-Scan system is designed to improve the patient experience with screening by eliminating many unattractive requirements, such as bowel preparation, fasting, and sedation.
Using ultra-low dose X-ray and wireless communication technologies, the capsule generates information on the contours of the inside of the colon as it passes naturally. This information is used to create a 3D map of the colon, which allows physicians to look for polyps and other abnormalities. Designed to improve the patient experience and increase the willingness of individuals to participate in recommended colorectal cancer screening, C-Scan removes many frequently-cited barriers, such as laxative bowel preparation, invasiveness and sedation. The C-Scan system is currently not cleared for marketing in any jurisdiction.
"The results of this critical phase of the collaboration are incredibly exciting for patients and the future prevention of colorectal cancer," said Emmanuel Ligner, general manager of Core Imaging for GE Healthcare Life Sciences. "GE Healthcare will continue its collaboration with Check-Cap using our expertise in radiopharmaceutical and device manufacturing to bring the C-Scan system to clinicians and patients."
Check-Cap is a clinical-stage medical diagnostics company developing C-Scan, the first capsule-based system for preparation-free colorectal cancer screening.
GE Healthcare provides transformational medical technologies and services to meet the demand for increased access, enhanced quality and more affordable healthcare around the world. From medical imaging, software & IT, patient monitoring and diagnostics to drug discovery, biopharmaceutical manufacturing technologies and performance improvement solutions, GE Healthcare helps medical professionals deliver healthcare to their patients.
Reference:
1. "Key Statistics for Colorectal Cancer," American Cancer Society, https://www.cancer.org/cancer/colon-rectal-cancer/about/key-statistics.html