Fortimedix Surgical B.V.10.18.16
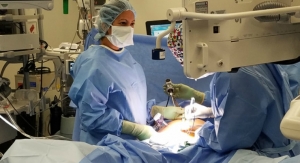
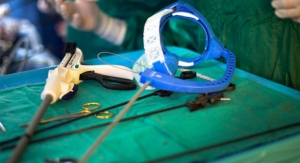
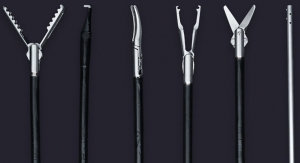
Fortimedix Surgical B.V. today officially launched in the United States the symphonX, a single-port surgery solution that is compatible with a standard 15 mm trocar for use in minimally invasive abdominal laparoscopic surgery.
“We are thrilled to formally launch the symphonX Surgical Platform in the U.S., which we believe has the potential to deliver on the promise of single-port surgery and elevate the standard of care in laparoscopy,” said Tom Dempsey, vice president of sales at Fortimedix Surgical. “The technology was developed under the engineering code name FMX314 in close collaboration with leading surgeons to ensure that the platform addresses the needs of the medical community, and by extension the patient community. Our early clinical experience underlines its potential for improved patient satisfaction and health outcomes, including fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis.”
symphonX addresses unmet needs in minimally invasive surgery by providing the medical community with a surgical solution that is small, simple and secure. Emulating conventional, multi-port laparoscopy, makes symphonX™ easy to use and enables surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
The first two U.S. commercial procedures with symphonX were recently performed at the University of California-San Diego Health by Santiago Horgan, M.D., following U.S. Food and Drug Administration 510(k) clearance and CE Mark approval in Europe. The European commercial launch is planned for 2017.
Fortimedix Surgical develops medical devices that capture the claimed benefits of single-port surgery. The company is headquartered in Nuth, The Netherlands, and has a U.S. subsidiary in San Diego, Calif.
“We are thrilled to formally launch the symphonX Surgical Platform in the U.S., which we believe has the potential to deliver on the promise of single-port surgery and elevate the standard of care in laparoscopy,” said Tom Dempsey, vice president of sales at Fortimedix Surgical. “The technology was developed under the engineering code name FMX314 in close collaboration with leading surgeons to ensure that the platform addresses the needs of the medical community, and by extension the patient community. Our early clinical experience underlines its potential for improved patient satisfaction and health outcomes, including fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis.”
symphonX addresses unmet needs in minimally invasive surgery by providing the medical community with a surgical solution that is small, simple and secure. Emulating conventional, multi-port laparoscopy, makes symphonX™ easy to use and enables surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
The first two U.S. commercial procedures with symphonX were recently performed at the University of California-San Diego Health by Santiago Horgan, M.D., following U.S. Food and Drug Administration 510(k) clearance and CE Mark approval in Europe. The European commercial launch is planned for 2017.
Fortimedix Surgical develops medical devices that capture the claimed benefits of single-port surgery. The company is headquartered in Nuth, The Netherlands, and has a U.S. subsidiary in San Diego, Calif.