Globe Newswire04.06.16
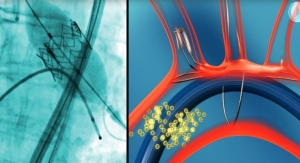
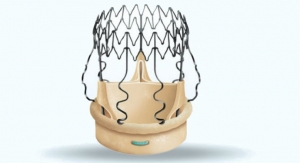
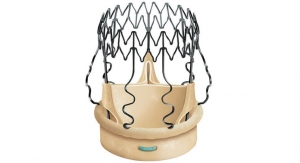
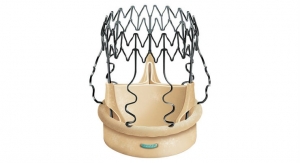
LivaNova plc announced the first implant of the Perceval sutureless valve in the PERSIST-AVR (Perceval Sutureless Implant Vs. Standard Aortic Valve Replacement) trial at CHU Brabois, University of Lorraine in Nancy, France. Engineered to restore the natural valve performance, Perceval is a sutureless heart valve for patients who require replacement of their aortic valve.
PERSIST-AVR is the first worldwide, prospective, randomized, multi-center trial comparing the Perceval sutureless aortic valve with standard sutured bioprostheses in patients with aortic valve disease. The study will enroll more than 1,200 patients with severe symptomatic aortic stenosis or steno-insufficiency in patients who are candidates for surgical replacement of their native aortic valve. The primary endpoint of the trial is non-inferiority of major adverse cardiac cerebrovascular events (MACCE) at one year according to VARC-2 criteria.1
"The clinical experience with the Perceval valve has been positive on multiple fronts as measured by reduced cross-clamp time, great hemodynamic performance, low structural valve deterioration and freedom from reoperation up to five-year follow-up.2 PERSIST-AVR is designed to confirm these results compared to standard sutured stented valves. Moreover, the trial will demonstrate how the use of Perceval in aortic valve surgery is linked with significant cost savings, driven primarily by reduced procedural costs and reduced hospital stay," said Professor Thierry Folliguet, M.D., Ph.D., who enrolled the first patient in PERSIST-AVR. Folliguet is part of the study's steering committee and one of the early implanters of Perceval.
Conducted worldwide, the study is planned to have a two-year enrollment period and a yearly follow-up for a five-year period. The primary endpoint is expected to be available in 2019.
"We know that PERSIST-AVR is a landmark clinical trial, being the first randomized study in 30 years in the field of valvular surgery. Its initiation is a great endeavor in the cardiac surgery communities. More than 60 centers around the world will be recruited pointing out the strong interest generated by Perceval. We look forward to obtaining promising data for the use of Perceval in the daily aortic valve replacement setting," said Professor Theodor Fischlein, M.D., Ph.D., of Paracelsus Medical University Cardiovascular Center in Nuremberg, Germany, and Dr. Roberto Lorusso, M.D., Ph.D., of the Cardio-Thoracic Surgery Department at Maastricht University Medical Centre (MUMC+) in Maastricht, The Netherlands, both principal investigators of the study.
To date, the Perceval sutureless valve has been implanted in more than 15,000 patients in more than 310 hospitals worldwide. More than 115 publications testify the good clinical results obtained with the device. The Perceval valve is approved for use in Europe, the United States and other international markets including Canada and Australia.
"As the first trial of its kind, the PERSIST-AVR study is a significant milestone for LivaNova, the healthcare community and patients worldwide," said Michel Darnaud, president of cardiac surgery B.U., LivaNova. "We are proud to support this trial. We anticipate its results could significantly impact daily practice and establish the Perceval sutureless valve as the prosthesis of choice for future heart valve surgeries."
LivaNova is a developer and manufacturer of medical devices used for cardiac surgery, neuromodulation and cardiac rhythm management. Formed by the merger of Sorin S.p.A. and Cyberonics Inc., the company employs roughly 4,500 people worldwide and has a presence in more than 100 countries. LivaNova operates three business units: Cardiac Rhythm Management, Cardiac Surgery, and Neuromodulation, and has operating headquarters in Clamart, France; Mirandola, Italy; and Houston, Texas.
References:
1. Kappetein, A. P., Head S. J., Genereux P., et al. "Updated Standardized Endpoint Definitions for Transcatheter Aortic Valve Implantation: The Valve Academic Research Consortium-2 Consensus Document." J Thorac Cardiovasc Surg 145, no. 1 (2013): 6-23.
2. Shrestha M. , Fischlein T., Meuris B. et al. "European multicentre experience with the sutureless Perceval valve: clinical and haemodynamic outcomes up to 5 years in over 700 patients" Eur J Cardiothorac Surg. 2016 Jan;49(1):234-41
PERSIST-AVR is the first worldwide, prospective, randomized, multi-center trial comparing the Perceval sutureless aortic valve with standard sutured bioprostheses in patients with aortic valve disease. The study will enroll more than 1,200 patients with severe symptomatic aortic stenosis or steno-insufficiency in patients who are candidates for surgical replacement of their native aortic valve. The primary endpoint of the trial is non-inferiority of major adverse cardiac cerebrovascular events (MACCE) at one year according to VARC-2 criteria.1
"The clinical experience with the Perceval valve has been positive on multiple fronts as measured by reduced cross-clamp time, great hemodynamic performance, low structural valve deterioration and freedom from reoperation up to five-year follow-up.2 PERSIST-AVR is designed to confirm these results compared to standard sutured stented valves. Moreover, the trial will demonstrate how the use of Perceval in aortic valve surgery is linked with significant cost savings, driven primarily by reduced procedural costs and reduced hospital stay," said Professor Thierry Folliguet, M.D., Ph.D., who enrolled the first patient in PERSIST-AVR. Folliguet is part of the study's steering committee and one of the early implanters of Perceval.
Conducted worldwide, the study is planned to have a two-year enrollment period and a yearly follow-up for a five-year period. The primary endpoint is expected to be available in 2019.
"We know that PERSIST-AVR is a landmark clinical trial, being the first randomized study in 30 years in the field of valvular surgery. Its initiation is a great endeavor in the cardiac surgery communities. More than 60 centers around the world will be recruited pointing out the strong interest generated by Perceval. We look forward to obtaining promising data for the use of Perceval in the daily aortic valve replacement setting," said Professor Theodor Fischlein, M.D., Ph.D., of Paracelsus Medical University Cardiovascular Center in Nuremberg, Germany, and Dr. Roberto Lorusso, M.D., Ph.D., of the Cardio-Thoracic Surgery Department at Maastricht University Medical Centre (MUMC+) in Maastricht, The Netherlands, both principal investigators of the study.
To date, the Perceval sutureless valve has been implanted in more than 15,000 patients in more than 310 hospitals worldwide. More than 115 publications testify the good clinical results obtained with the device. The Perceval valve is approved for use in Europe, the United States and other international markets including Canada and Australia.
"As the first trial of its kind, the PERSIST-AVR study is a significant milestone for LivaNova, the healthcare community and patients worldwide," said Michel Darnaud, president of cardiac surgery B.U., LivaNova. "We are proud to support this trial. We anticipate its results could significantly impact daily practice and establish the Perceval sutureless valve as the prosthesis of choice for future heart valve surgeries."
LivaNova is a developer and manufacturer of medical devices used for cardiac surgery, neuromodulation and cardiac rhythm management. Formed by the merger of Sorin S.p.A. and Cyberonics Inc., the company employs roughly 4,500 people worldwide and has a presence in more than 100 countries. LivaNova operates three business units: Cardiac Rhythm Management, Cardiac Surgery, and Neuromodulation, and has operating headquarters in Clamart, France; Mirandola, Italy; and Houston, Texas.
References:
1. Kappetein, A. P., Head S. J., Genereux P., et al. "Updated Standardized Endpoint Definitions for Transcatheter Aortic Valve Implantation: The Valve Academic Research Consortium-2 Consensus Document." J Thorac Cardiovasc Surg 145, no. 1 (2013): 6-23.
2. Shrestha M. , Fischlein T., Meuris B. et al. "European multicentre experience with the sutureless Perceval valve: clinical and haemodynamic outcomes up to 5 years in over 700 patients" Eur J Cardiothorac Surg. 2016 Jan;49(1):234-41