Nemaura Medical Inc.03.15.16
Loughborough, England-based Nemaura Medical Inc. a medical device company developing a minimally invasive wireless continuous glucose monitoring (CGM) system known as Sugarbeat, announced today that it has received notification of CE mark approval for its Sugarbeat system. The device for which CE grant notice has been approved was used in a 160 patient day study generating over 1,500 paired data points, and retrospective evaluation of the glucose data. Nemaura expects the CE certificate to be received in April, upon completion of its 3-yearly ISO13485 quality systems recertification process, for which it was audited at the end of February this year.
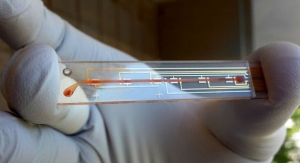
The Sugarbeat CGM system is a body worn sensor patch designed to display real time glucose readings on a standalone reader, or smart watch or smart phone app via blue tooth, or allow retrospective evaluation of the data for glucose trending, as an adjunct device. A comprehensive 10 year study, The Diabetes Control and Complications Trial, funded by the National Institute of Diabetes and Digestive and Kidney Disease, showed that maintaining better control over blood sugar levels delayed the onset and progression of complications of diabetes, including diseases of the eye, kidney and nerve damage, caused by diabetes.
Nemaura’s system is expected to empower patients to gain better control over their blood sugar levels using the minimally-invasive Sugarbeat system, which is expected to be cost efficient compared to current marketed CGM’s as well as allow the user to decide when to wear the patch, and not have to retain the patch on the skin permanently. Nemaura are quoted as saying ‘one of the key reasons people have cited for not using current CGM’s, apart from cost, is their invasive nature and the need to retain the device on the body for long durations of up to 14 days, even at times when the patient would otherwise choose not to wear the device.
Nemaura Medical is currently in the process of completing a second pre-submission to the U.S. Food and Drug Administration to discuss their clinical strategy for the pre-market approval route, and expects to commence U.S. studies by mid 2016. The European launch of the Sugarbeat is anticipated to be by the end of 2016, with their European joint venture partner, DBJ Ltd.
The Sugarbeat CGM system is a body worn sensor patch designed to display real time glucose readings on a standalone reader, or smart watch or smart phone app via blue tooth, or allow retrospective evaluation of the data for glucose trending, as an adjunct device. A comprehensive 10 year study, The Diabetes Control and Complications Trial, funded by the National Institute of Diabetes and Digestive and Kidney Disease, showed that maintaining better control over blood sugar levels delayed the onset and progression of complications of diabetes, including diseases of the eye, kidney and nerve damage, caused by diabetes.
Nemaura’s system is expected to empower patients to gain better control over their blood sugar levels using the minimally-invasive Sugarbeat system, which is expected to be cost efficient compared to current marketed CGM’s as well as allow the user to decide when to wear the patch, and not have to retain the patch on the skin permanently. Nemaura are quoted as saying ‘one of the key reasons people have cited for not using current CGM’s, apart from cost, is their invasive nature and the need to retain the device on the body for long durations of up to 14 days, even at times when the patient would otherwise choose not to wear the device.
Nemaura Medical is currently in the process of completing a second pre-submission to the U.S. Food and Drug Administration to discuss their clinical strategy for the pre-market approval route, and expects to commence U.S. studies by mid 2016. The European launch of the Sugarbeat is anticipated to be by the end of 2016, with their European joint venture partner, DBJ Ltd.