CBSET12.17.15
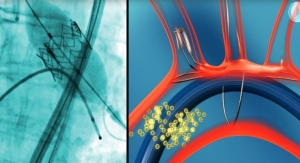
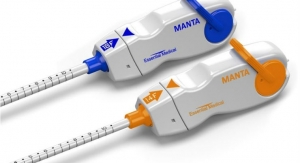
Scientists at CBSET Inc., a not-for-profit preclinical research institute, have presented preclinical evidence that the MANTA Large Bore Vascular Closure Device (VCD) from Essential Medical (Malvern, Pa.) successfully closed large bore arterial access without complication and exhibited favorable healing characteristics as assessed by serial histomorphology.
Vascular access site complications are a significant source of morbidity following percutaneous catheterization, especially for interventions requiring large bore cannulation, such as transcatheter aortic valve replacement (TAVR), causing a long time to achieve hemostasis and extending operating room time, requiring the need for a vascular surgeon in the cath lab, delaying patient ambulation, causing a high rate of complications and high total cost of care.
“The benefit of a vascular closure device over the standard of manual compression is most pronounced for large bore access and higher baseline-risk patients. The fact that the MANTA VCD uses a collagen implant secured by an intraluminal anchor makes it especially suited for large bore cannulation,” said Haim Danenberg, M.D., chief scientific advisor at CBSET, and head of the Cardiovascular Research Center and director of Interventional Cardiology at the Heart Institute of Hadassah Hebrew University Medical Center in Jerusalem, Israel.
In July 2015, Essential Medical announced that it had begun European clinical studies using its MANTA Large Bore Vascular Closure device, designed to seal both 14F and 18F large bore femoral punctures. Cases were conducted by Nicholas Van Mieghem, M.D., Ph.D., medical director of the Department of Interventional Cardiology at the Thoraxcenter, Erasmus Medical Center in The Netherlands, who called MANTA, a "revolutionary device" that will transform how doctors close cases.
“MANTA is designed to close a large arteriotomy left by vascular access sheaths in high-pressure vessels in less than 60 seconds, providing immediate hemostasis in order to reduce complications associated with large bore closure,” said study director Lynn Bailey, director of Interventional Services at CBSET.
“In our study, the MANTA VCD rapidly achieved patent hemostasis, and there was no evidence of vascular occlusion or other complications on long-term follow-up,” added lead author Kenta Nakamura, M.D., fellow in Cardiovascular Disease at Massachusetts General Hospital, and research fellow in Applied Sciences at CBSET. “The device was well-tolerated with favorable cellular response and tissue healing on histopathology.”
“With CBSET’s help, Essential Medical has been able to cost-effectively validate its disruptive technology by developing and employing the most appropriate animal models. We are excited to be able to couple our clinical science with preclinical science,” said Gary Roubin, M.D., Ph.D., chief medical officer at Essential Medical.
Based in Lexington, Mass., CBSET bills itself as a preclinical research leader in critically important therapeutic fields such as interventional cardiology, renal disease and dialysis, chronic drug-resistant hypertension, women’s health, minimally invasive surgery, orthopedics, biological and synthetic tissue repair, drug delivery, bioresorbable devices, and combination medical device and drug-eluting products.
Vascular access site complications are a significant source of morbidity following percutaneous catheterization, especially for interventions requiring large bore cannulation, such as transcatheter aortic valve replacement (TAVR), causing a long time to achieve hemostasis and extending operating room time, requiring the need for a vascular surgeon in the cath lab, delaying patient ambulation, causing a high rate of complications and high total cost of care.
“The benefit of a vascular closure device over the standard of manual compression is most pronounced for large bore access and higher baseline-risk patients. The fact that the MANTA VCD uses a collagen implant secured by an intraluminal anchor makes it especially suited for large bore cannulation,” said Haim Danenberg, M.D., chief scientific advisor at CBSET, and head of the Cardiovascular Research Center and director of Interventional Cardiology at the Heart Institute of Hadassah Hebrew University Medical Center in Jerusalem, Israel.
In July 2015, Essential Medical announced that it had begun European clinical studies using its MANTA Large Bore Vascular Closure device, designed to seal both 14F and 18F large bore femoral punctures. Cases were conducted by Nicholas Van Mieghem, M.D., Ph.D., medical director of the Department of Interventional Cardiology at the Thoraxcenter, Erasmus Medical Center in The Netherlands, who called MANTA, a "revolutionary device" that will transform how doctors close cases.
“MANTA is designed to close a large arteriotomy left by vascular access sheaths in high-pressure vessels in less than 60 seconds, providing immediate hemostasis in order to reduce complications associated with large bore closure,” said study director Lynn Bailey, director of Interventional Services at CBSET.
“In our study, the MANTA VCD rapidly achieved patent hemostasis, and there was no evidence of vascular occlusion or other complications on long-term follow-up,” added lead author Kenta Nakamura, M.D., fellow in Cardiovascular Disease at Massachusetts General Hospital, and research fellow in Applied Sciences at CBSET. “The device was well-tolerated with favorable cellular response and tissue healing on histopathology.”
“With CBSET’s help, Essential Medical has been able to cost-effectively validate its disruptive technology by developing and employing the most appropriate animal models. We are excited to be able to couple our clinical science with preclinical science,” said Gary Roubin, M.D., Ph.D., chief medical officer at Essential Medical.
Based in Lexington, Mass., CBSET bills itself as a preclinical research leader in critically important therapeutic fields such as interventional cardiology, renal disease and dialysis, chronic drug-resistant hypertension, women’s health, minimally invasive surgery, orthopedics, biological and synthetic tissue repair, drug delivery, bioresorbable devices, and combination medical device and drug-eluting products.