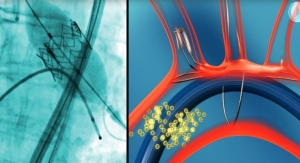
X-Seal closes femoral arterial punctures made during cardiac catheterization procedures such as angiograms, angioplasty, and stenting. X-Seal is designed to provide Cath Lab professionals with superior closure and improved hemostasis, according to the company. The potential worldwide market for vascular closure products is approximately $1 billion.
"X-Seal represents a truly 'next-generation' closure device, offering excellent sealing, angiographic marking, and controlled deployment," said Gary Roubin, M.D., Ph.D., an interventional cardiologist and co-founder of Essential Medical.
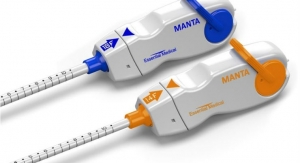
In addition to X-Seal, Essential Medical is developing Manta, a large bore device for sealing larger femoral arterial punctures made during transcatheter aortic valve replacement (TAVR), balloon aortic valvuloplasty (BAV), and the endovascular treatment of abdominal aortic aneurysms (AAA). The large bore device uses sealing components similar to X-Seal, and can be deployed within 60 seconds. First in man studies are starting soon and a larger EU study is expected to begin in 2015. "We expect our large bore device will provide a closure solution that will improve patient outcomes and reduce the overall cost of the respective procedures with an easy to use product," CEO Greg Walters said.
Based in Malvern, Pa., Essential Medical develops and commercializes new vascular closure devices for femoral punctures after diagnostic and interventional cardiac catheterization procedures. The company was founded in 2010 by Walters, a senior product development executive with 20 years' experience creating innovative cardiovascular products, and Roubin, a coronary and carotid stenting physician.