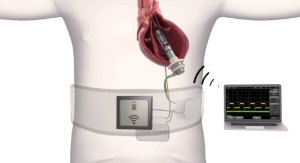
CardiacAssist Inc.'s Protek Duo emergency cannula has been granted CE Mark approval.
The device uses a minimally invasive percutaneous process to administer veno-venous extracorporeal life support for patients with acute respiratory disease syndrome. The cannula is inserted into the jugular vein and can both drain and re-infuse blood, the company said.
Health professionals can use the Protek Duo in conjunction with Cardiac Assist’s TandemHeart life support system, which provides up to 30 days of circulatory support, the company said.
Cardiac Assist received U.S. Food and Drug Administration (FDA) approval for the Protek Duo in August 2014.
In May 2014, the FDA issued a warning letter against Cardiac Assist, claiming the devicemaker did not report the death of a patient using the TandemHeart pump. The FDA deemed the company’s response sufficient and the matter was resolved.
CardiacAssist Inc. is a Pittsburgh, Pa.-based company founded in 1996 that develops, manufactures and markets circulatory support and other cardiovascular systems for cardiologists and cardiac surgeons.