07.03.15
Mountain View, Calif.-based Hansen Medical Inc., which makes intravascular robotics, has completed enrollment in the initial evaluable cohort of the Artisan clinical trial.
The Artisan atrial fibrillation (AF) trial is a prospective, single-arm, multi-center, U.S. and outside U.S. study designed to evaluate the safety and effectiveness of the Sensei robotic system and Artisan family of catheters for introducing and positioning specified commercially available RF (radiofrequency) ablation catheters in subjects with symptomatic, drug-refractory paroxysmal atrial fibrillation. The Bayesian designed study requires enrollment of an initial cohort of 125 patients, with subsequent enrollment permitted to support the end-point analysis. The trial has two primary endpoints: safety, defined as the absence of early onset of all major adverse events, and efficacy, defined as freedom from atrial fibrillation through one year.
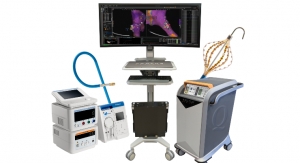
The Sensei robotic system combines advanced levels of 3-D catheter control and 3-D visualization. technology is powered by a robotically controlled arm designed for catheter navigation, stability and positioning within the patient’s heart atria. The Sensei robotic system, control catheters and accessories are intended to facilitate manipulation, positioning and control of Hansen Medical’s robotically steerable catheters for collecting electrophysiological data within the heart atria with electro-anatomic mapping and recording systems, using specified percutaneous mapping catheters. The system is powered by a robotically controlled arm meant to allow for catheter navigation and stability. The safety and effectiveness of this device for use with cardiac ablation catheters, in the treatment of cardiac arrhythmias including atrial fibrillation, have not been established.
“We are excited to see the initial phase of enrollment completed in the [investigational device excemption trial] and are working closely with the investigators and U.S. Food and Drug Administration to expand the clinical indications for the Artisan catheter in the U.S. market,” said Cary Vance, president and CEO of Hansen Medical. “This patient cohort sets the initial timing to evaluate the trial endpoints and gain additional clinical indications in the United States. We believe our robotic technology brings enhanced control and predictability to these procedures, and addresses serious occupational hazards faced by physicians and their teams in the delivery of patient care.”
“This clinical trial is significant and sets a milestone for robotic technology in the treatment of patients with atrial fibrillation,” said Joseph Gallinghouse, M.D., of the Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin, Texas. “The team here at St. David’s finds the Sensei Robotic System reduces radiation exposure to the operator, while the Artisan catheter predictably navigates and provides a stable platform during AF ablation, a procedure which can sometimes last for hours.”
Gallinghouse has significant experience with the Sensei Robotic System and Artisan catheter and has performed more than 1,200 procedures to date.
Cardiac arrhythmias are abnormal electrical signals in the heart. Atrial fibrillation is the most common form of cardiac arrhythmia, affecting nearly 3 million people in the United States alone. In radiofrequency catheter ablation, a catheter is inserted into the left atrium and radiofrequency energy is delivered to the heart tissue to create scars, which are intended to block erratic electrical impulses so the left atrium can beat normally.
The Artisan atrial fibrillation (AF) trial is a prospective, single-arm, multi-center, U.S. and outside U.S. study designed to evaluate the safety and effectiveness of the Sensei robotic system and Artisan family of catheters for introducing and positioning specified commercially available RF (radiofrequency) ablation catheters in subjects with symptomatic, drug-refractory paroxysmal atrial fibrillation. The Bayesian designed study requires enrollment of an initial cohort of 125 patients, with subsequent enrollment permitted to support the end-point analysis. The trial has two primary endpoints: safety, defined as the absence of early onset of all major adverse events, and efficacy, defined as freedom from atrial fibrillation through one year.
The Sensei robotic system combines advanced levels of 3-D catheter control and 3-D visualization. technology is powered by a robotically controlled arm designed for catheter navigation, stability and positioning within the patient’s heart atria. The Sensei robotic system, control catheters and accessories are intended to facilitate manipulation, positioning and control of Hansen Medical’s robotically steerable catheters for collecting electrophysiological data within the heart atria with electro-anatomic mapping and recording systems, using specified percutaneous mapping catheters. The system is powered by a robotically controlled arm meant to allow for catheter navigation and stability. The safety and effectiveness of this device for use with cardiac ablation catheters, in the treatment of cardiac arrhythmias including atrial fibrillation, have not been established.
“We are excited to see the initial phase of enrollment completed in the [investigational device excemption trial] and are working closely with the investigators and U.S. Food and Drug Administration to expand the clinical indications for the Artisan catheter in the U.S. market,” said Cary Vance, president and CEO of Hansen Medical. “This patient cohort sets the initial timing to evaluate the trial endpoints and gain additional clinical indications in the United States. We believe our robotic technology brings enhanced control and predictability to these procedures, and addresses serious occupational hazards faced by physicians and their teams in the delivery of patient care.”
“This clinical trial is significant and sets a milestone for robotic technology in the treatment of patients with atrial fibrillation,” said Joseph Gallinghouse, M.D., of the Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin, Texas. “The team here at St. David’s finds the Sensei Robotic System reduces radiation exposure to the operator, while the Artisan catheter predictably navigates and provides a stable platform during AF ablation, a procedure which can sometimes last for hours.”
Gallinghouse has significant experience with the Sensei Robotic System and Artisan catheter and has performed more than 1,200 procedures to date.
Cardiac arrhythmias are abnormal electrical signals in the heart. Atrial fibrillation is the most common form of cardiac arrhythmia, affecting nearly 3 million people in the United States alone. In radiofrequency catheter ablation, a catheter is inserted into the left atrium and radiofrequency energy is delivered to the heart tissue to create scars, which are intended to block erratic electrical impulses so the left atrium can beat normally.