07.02.15
GlySure Ltd., a medical device firm based in Abingdon, England, has received CE mark for what the company claims is the world’s “first and only continuous intravascular glucose monitoring system (CIGMS).”
The initial use of the GlySure CIGMS is to enhance blood glucose management among adult cardiac surgery patients in the intensive care unit (ICU). The company currently is conducting a United Kingdom-based multi-center trial designed to enable use of the GlySure CIGMS across all adult intensive care patients.
In critically ill patients, poorly controlled blood sugar levels can lead to increased mortality, morbidity, lengths of stay in the ICU and costs to healthcare providers. The company plans to first bring its technology to hospitals in the United Kingdom, Benelux countries and Germany, followed by launches in additional European countries and other markets where CE mark is recognized.
“For over a decade, the clinical community has been seeking a way to tightly control glucose levels in critically ill patients for both improved outcomes and reduced costs,” said Krishna Prasad, M.D., consultant anesthesiologist at Care Hospitals, Nampally, in Hyderabad, India, who was the principal investigator of the CIGMS CE marking trial. “GlySure’s technology enables them to do this safely with accuracy, reliability and efficiency to support the implementation of improved glycemic control protocols.”
“We are pleased to bring to market the first practical solution to address this significant unmet medical need,” said Roger Moody, chief operating officer for GlySure. “Our first customers will lead the way in how glucose management is best practiced, enabling improved patient outcomes and reduced healthcare costs. Continuous glucose monitoring in critical care has been a vexing medical challenge. GlySure’s game changing technology and elegant CIGMS solution will be the first in this market to deliver clinical value to patients and savings to healthcare providers.”
GlySure received its CE Mark following a review of its submission that included the results of its multi-center clinical trial that demonstrated the system’s ability to provide accurate continuous blood glucose monitoring in patients throughout their length of ICU stay. The GlySure CIGMS met the primary safety and efficacy endpoints and demonstrated a consistently high level of accuracy compared to a “gold standard” intermittent glucose analyzer.
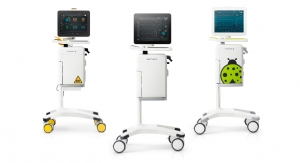
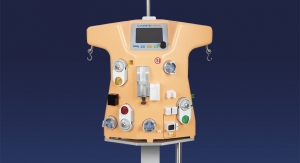
The GlySure CIGMS comprises three main parts: a monitor, a disposable fiber optic sensor and a disposable five-lumen central venous catheter, similar to that typically used in the ICU. According to the company, the GlySure sensor includes a highly selective proprietary chemistry that provides the first commercially available glucose testing system that can accurately measure intravascular glucose levels every 15 seconds. This provides physicians with continual feedback that helps avoid dangerous fluctuations in ICU patient glucose levels.
GlySure intends to seek clearance from the U.S. Food and Drug Administration for the GlySure CIGMS and is currently finalizing the design of the clinical trial to support its submission. The company recently appointed Albert Leung, M.D., Ph.D., as its chief medical officer to lead these clinical and regulatory efforts. Leung is an endocrinologist with 20 years of clinical practice and industry experience, and had previously worked at Merck and Johnson & Johnson to bring new products to market by leading their clinical development programs.
GlySure estimates the global market opportunity for continuous glucose monitoring in the ICU is greater than $5 billion. This calculation takes into consideration the total number of patients in a critical care setting worldwide.
Recent research has demonstrated that controlling patient glucose levels in the ICU within tight normal ranges yielded significant improvements in patient outcomes, including a 46 percent reduction in incidence of sepsis, 41 percent reduction in renal failure, 50 percent reduction in blood transfusions and 34 percent reduction in mortality, according to data cited by the company. This study created a new field of medical research with over 100 publications in the past decade including two showing a financial benefit to the hospital in savings of $1,580 and 2,638 British pounds per patient, respectively.1,2 More recently, the NICE-SUGAR trial provides evidence that patients have better outcomes when glucose levels are higher and hypoglycemia events are lower.3
References
1. Krinsley JS, Jones RL. Cost analysis of intensive glycemic control in critically ill adult patients. Chest. 129(3), 644-650 (2006).
2. Van den Berghe G, et al. Analysis of healthcare resource utilization with intensive insulin therapy in critically ill patients. Crit Care Med. 34(3), 612-6 (2006).
3. The NICE-SUGAR Study Investigators. Hypoglycemia and risk of death in critically ill patients. N Engl J Med. 367, 1108-1118 (2012).
The initial use of the GlySure CIGMS is to enhance blood glucose management among adult cardiac surgery patients in the intensive care unit (ICU). The company currently is conducting a United Kingdom-based multi-center trial designed to enable use of the GlySure CIGMS across all adult intensive care patients.
In critically ill patients, poorly controlled blood sugar levels can lead to increased mortality, morbidity, lengths of stay in the ICU and costs to healthcare providers. The company plans to first bring its technology to hospitals in the United Kingdom, Benelux countries and Germany, followed by launches in additional European countries and other markets where CE mark is recognized.
“For over a decade, the clinical community has been seeking a way to tightly control glucose levels in critically ill patients for both improved outcomes and reduced costs,” said Krishna Prasad, M.D., consultant anesthesiologist at Care Hospitals, Nampally, in Hyderabad, India, who was the principal investigator of the CIGMS CE marking trial. “GlySure’s technology enables them to do this safely with accuracy, reliability and efficiency to support the implementation of improved glycemic control protocols.”
“We are pleased to bring to market the first practical solution to address this significant unmet medical need,” said Roger Moody, chief operating officer for GlySure. “Our first customers will lead the way in how glucose management is best practiced, enabling improved patient outcomes and reduced healthcare costs. Continuous glucose monitoring in critical care has been a vexing medical challenge. GlySure’s game changing technology and elegant CIGMS solution will be the first in this market to deliver clinical value to patients and savings to healthcare providers.”
GlySure received its CE Mark following a review of its submission that included the results of its multi-center clinical trial that demonstrated the system’s ability to provide accurate continuous blood glucose monitoring in patients throughout their length of ICU stay. The GlySure CIGMS met the primary safety and efficacy endpoints and demonstrated a consistently high level of accuracy compared to a “gold standard” intermittent glucose analyzer.
The GlySure CIGMS comprises three main parts: a monitor, a disposable fiber optic sensor and a disposable five-lumen central venous catheter, similar to that typically used in the ICU. According to the company, the GlySure sensor includes a highly selective proprietary chemistry that provides the first commercially available glucose testing system that can accurately measure intravascular glucose levels every 15 seconds. This provides physicians with continual feedback that helps avoid dangerous fluctuations in ICU patient glucose levels.
GlySure intends to seek clearance from the U.S. Food and Drug Administration for the GlySure CIGMS and is currently finalizing the design of the clinical trial to support its submission. The company recently appointed Albert Leung, M.D., Ph.D., as its chief medical officer to lead these clinical and regulatory efforts. Leung is an endocrinologist with 20 years of clinical practice and industry experience, and had previously worked at Merck and Johnson & Johnson to bring new products to market by leading their clinical development programs.
GlySure estimates the global market opportunity for continuous glucose monitoring in the ICU is greater than $5 billion. This calculation takes into consideration the total number of patients in a critical care setting worldwide.
Recent research has demonstrated that controlling patient glucose levels in the ICU within tight normal ranges yielded significant improvements in patient outcomes, including a 46 percent reduction in incidence of sepsis, 41 percent reduction in renal failure, 50 percent reduction in blood transfusions and 34 percent reduction in mortality, according to data cited by the company. This study created a new field of medical research with over 100 publications in the past decade including two showing a financial benefit to the hospital in savings of $1,580 and 2,638 British pounds per patient, respectively.1,2 More recently, the NICE-SUGAR trial provides evidence that patients have better outcomes when glucose levels are higher and hypoglycemia events are lower.3
References
1. Krinsley JS, Jones RL. Cost analysis of intensive glycemic control in critically ill adult patients. Chest. 129(3), 644-650 (2006).
2. Van den Berghe G, et al. Analysis of healthcare resource utilization with intensive insulin therapy in critically ill patients. Crit Care Med. 34(3), 612-6 (2006).
3. The NICE-SUGAR Study Investigators. Hypoglycemia and risk of death in critically ill patients. N Engl J Med. 367, 1108-1118 (2012).