06.24.15
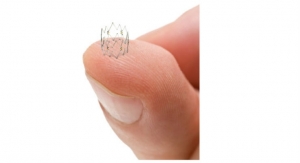
Spectranetics Corp. received U.S. Food and Drug Administration 510(k) clearance for the AngioSculpt PTA Scoring Balloon Catheter in 7 and 8 millimeter (mm) diameter balloon sizes.
According to officials from the Colorado Springs, Colo.-based firm, the products are designed to treat the narrowing of the native or synthetic fistula, a surgically created conduit between an artery and vein used to remove and return blood during hemodialysis.
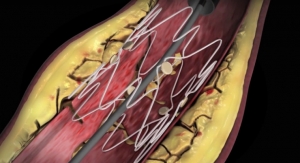
Approximately half of all patients who undergo balloon angioplasty will return with recurrent hemodialysis access dysfunction within six months, according to figures cited by the company. The AngioSculpt PTA Scoring Balloon Catheter in 7 and 8 mm diameter assists physicians with a difficult patient set, targeting lesions in the hemodialysis access that are resistant to standard balloon angioplasty.
"In spite of advances in medical device technology, maintaining hemodialysis access function remains a difficult challenge," said George Mueller, M.D., with Bethesda Hospital in Boynton Beach, Fla. "The AngioSculpt Scoring Balloon Catheter, now in sizes appropriate for a larger range of vessel diameters, has the potential to address the most difficult problems we encounter in treating AV (arteriovenous) access dysfunction. The product provides the power physicians need, partnered with a controlled dilatation that reduces dissection rates and trauma to the vessel. Physicians who frequently treat these patients are hopeful that adoption of the new tool will demonstrate that it not only preserves, but extends the life of an access."
In addition to the hemodialysis access market, the AngioSculpt Scoring Balloon Catheter in 7 and 8 mm diameter also is appropriately sized to prepare highly calcified lesions in the iliac and superficial femoral arteries. These lesions traditionally challenge routine stent placement. Due to the product's lower inflation pressures, the larger diameter AngioSculpt provides a safe approach to preparing the vessel for optimal stent placement, company officials noted.
"This is a large market with ample opportunity to improve patient outcomes with superior technology, combining precision, power and safety," said Kim Bridges, senior vice president of sales and marketing, Vascular Intervention, Spectranetics. "The 7 and 8 mm AngioSculpt expands the AngioSculpt product line, the number one specialty balloon in the market, and extends Spectranetics' comprehensive portfolio to cross, prepare and treat the most complex cardiovascular conditions, now to include hemodialysis access."
AngioSculpt Scoring Balloon Catheters treat coronary and peripheral artery disease. The devices have been used in more than 350,000 procedures worldwide, according to the company.
Spectranetics makes medical devices used in minimally invasive procedures within the cardiovascular system. The company's vascular intervention products include a range of laser catheters for ablation of blockages in arteries above and below the knee, the AngioSculpt scoring balloon used in peripheral and coronary procedures and Stellarex drug-coated balloon peripheral angioplasty platform, which received European CE mark approval in December 2014. The company also markets support catheters to facilitate crossing of peripheral and coronary arterial blockages, and retrograde access and guidewire retrieval devices used in the treatment of peripheral arterial blockages, including chronic total occlusions. The company markets aspiration and cardiac laser catheters to treat blockages in the heart. The Lead Management product line includes excimer laser sheaths, dilator sheaths, mechanical sheaths and accessories for the removal of pacemaker and defibrillator cardiac leads.
According to officials from the Colorado Springs, Colo.-based firm, the products are designed to treat the narrowing of the native or synthetic fistula, a surgically created conduit between an artery and vein used to remove and return blood during hemodialysis.
Approximately half of all patients who undergo balloon angioplasty will return with recurrent hemodialysis access dysfunction within six months, according to figures cited by the company. The AngioSculpt PTA Scoring Balloon Catheter in 7 and 8 mm diameter assists physicians with a difficult patient set, targeting lesions in the hemodialysis access that are resistant to standard balloon angioplasty.
"In spite of advances in medical device technology, maintaining hemodialysis access function remains a difficult challenge," said George Mueller, M.D., with Bethesda Hospital in Boynton Beach, Fla. "The AngioSculpt Scoring Balloon Catheter, now in sizes appropriate for a larger range of vessel diameters, has the potential to address the most difficult problems we encounter in treating AV (arteriovenous) access dysfunction. The product provides the power physicians need, partnered with a controlled dilatation that reduces dissection rates and trauma to the vessel. Physicians who frequently treat these patients are hopeful that adoption of the new tool will demonstrate that it not only preserves, but extends the life of an access."
In addition to the hemodialysis access market, the AngioSculpt Scoring Balloon Catheter in 7 and 8 mm diameter also is appropriately sized to prepare highly calcified lesions in the iliac and superficial femoral arteries. These lesions traditionally challenge routine stent placement. Due to the product's lower inflation pressures, the larger diameter AngioSculpt provides a safe approach to preparing the vessel for optimal stent placement, company officials noted.
"This is a large market with ample opportunity to improve patient outcomes with superior technology, combining precision, power and safety," said Kim Bridges, senior vice president of sales and marketing, Vascular Intervention, Spectranetics. "The 7 and 8 mm AngioSculpt expands the AngioSculpt product line, the number one specialty balloon in the market, and extends Spectranetics' comprehensive portfolio to cross, prepare and treat the most complex cardiovascular conditions, now to include hemodialysis access."
AngioSculpt Scoring Balloon Catheters treat coronary and peripheral artery disease. The devices have been used in more than 350,000 procedures worldwide, according to the company.
Spectranetics makes medical devices used in minimally invasive procedures within the cardiovascular system. The company's vascular intervention products include a range of laser catheters for ablation of blockages in arteries above and below the knee, the AngioSculpt scoring balloon used in peripheral and coronary procedures and Stellarex drug-coated balloon peripheral angioplasty platform, which received European CE mark approval in December 2014. The company also markets support catheters to facilitate crossing of peripheral and coronary arterial blockages, and retrograde access and guidewire retrieval devices used in the treatment of peripheral arterial blockages, including chronic total occlusions. The company markets aspiration and cardiac laser catheters to treat blockages in the heart. The Lead Management product line includes excimer laser sheaths, dilator sheaths, mechanical sheaths and accessories for the removal of pacemaker and defibrillator cardiac leads.