06.08.15
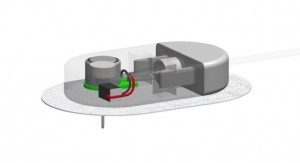
Franklin Lake, N.J.-based BD Medical, a segment of (Becton, Dickinson and Company) and Dublin, Ireland-based Medtronic plc are collaborating to introduce a new insulin pump infusion set with BD Flowsmart technology. An insulin infusion set delivers insulin from the pump to the body and is a critical part of the overall pump system. As part of the agreement, Medtronic will commercialize the infusion set, while BD will manufacture the product.
The infusion set has received both U.S. Food and Drugv Administration 510(k) clearance in April and Health Canada approval in January, and its launch is expected in 2016. An application has been submitted for the CE mark sun Europe. It is designed to work with both Medtronic insulin pumps and most other manufacturers’ insulin pumps.
“We are very excited to partner with a global leader like Medtronic to ensure that the benefits of
BD Flowsmart technology can be made widely available to as many people living with diabetes as possible,” said Kenneth Miller , worldwide president, BD Medical-Diabetes Care. “By increasing the reach and availability of this technology, we have an opportunity to enhance the use of insulin pump therapy by providing potentially more consistent insulin delivery and fewer flow interruptions. This technology is an extension of BD’s long-standing commitment to deliver injection solutions, and builds on previous BD research that has advanced the science and understanding of insulin delivery as well as patient comfort.”
“Medtronic is committed to advancing innovation across the entire insulin pump system, including infusion sets, to improve outcomes and expand options to satisfy different patient needs, body types and lifestyles,” said Annette Bruls, vice president and general manager, Diabetes Service and Solutions at Medtronic. “Our partnering with diabetes technology leaders, such as BD, will give more people with diabetes access to these innovations so they can enjoy greater freedom and better health.”
The insulin infusion set with BD Flowsmart technology was developed in collaboration with the Juvenile Diabetes Research Foundation (JDRF) and The Leona M. and Harry B. Helmsley Charitable Trust to improve the treatment of type 1 diabetes.
“JDRF is thrilled to have partnered with BD and the Helmsley Charitable Trust to bring the insulin infusion set with BD FlowSmart technology to people with type 1 diabetes,” said Aaron Kowalski , Ph.D., chief mission officer for JDRF. “This new infusion set is an important step forward in improving insulin pump therapy, and we hope ultimately, artificial pancreas systems.”
The insulin infusion set with BD FlowSmart technology features a side-ported catheter designed to improve insulin flow, potentially reducing the number of flow interruptions (defined as a continuous rise in inline pressure lasting at least 30 minutes). Without triggering a pump occlusion alarm, these events are known as silent occlusions, and because pump users may be unaware of these lapses in insulin flow, they could lead to unexplained hyperglycemia in some people on pump therapy. The infusion set also features a 30 gauge insertion needle, designed in such a small size so as to reduce insertion pain.
The infusion set has received both U.S. Food and Drugv Administration 510(k) clearance in April and Health Canada approval in January, and its launch is expected in 2016. An application has been submitted for the CE mark sun Europe. It is designed to work with both Medtronic insulin pumps and most other manufacturers’ insulin pumps.
“We are very excited to partner with a global leader like Medtronic to ensure that the benefits of
BD Flowsmart technology can be made widely available to as many people living with diabetes as possible,” said Kenneth Miller , worldwide president, BD Medical-Diabetes Care. “By increasing the reach and availability of this technology, we have an opportunity to enhance the use of insulin pump therapy by providing potentially more consistent insulin delivery and fewer flow interruptions. This technology is an extension of BD’s long-standing commitment to deliver injection solutions, and builds on previous BD research that has advanced the science and understanding of insulin delivery as well as patient comfort.”
“Medtronic is committed to advancing innovation across the entire insulin pump system, including infusion sets, to improve outcomes and expand options to satisfy different patient needs, body types and lifestyles,” said Annette Bruls, vice president and general manager, Diabetes Service and Solutions at Medtronic. “Our partnering with diabetes technology leaders, such as BD, will give more people with diabetes access to these innovations so they can enjoy greater freedom and better health.”
The insulin infusion set with BD Flowsmart technology was developed in collaboration with the Juvenile Diabetes Research Foundation (JDRF) and The Leona M. and Harry B. Helmsley Charitable Trust to improve the treatment of type 1 diabetes.
“JDRF is thrilled to have partnered with BD and the Helmsley Charitable Trust to bring the insulin infusion set with BD FlowSmart technology to people with type 1 diabetes,” said Aaron Kowalski , Ph.D., chief mission officer for JDRF. “This new infusion set is an important step forward in improving insulin pump therapy, and we hope ultimately, artificial pancreas systems.”
The insulin infusion set with BD FlowSmart technology features a side-ported catheter designed to improve insulin flow, potentially reducing the number of flow interruptions (defined as a continuous rise in inline pressure lasting at least 30 minutes). Without triggering a pump occlusion alarm, these events are known as silent occlusions, and because pump users may be unaware of these lapses in insulin flow, they could lead to unexplained hyperglycemia in some people on pump therapy. The infusion set also features a 30 gauge insertion needle, designed in such a small size so as to reduce insertion pain.