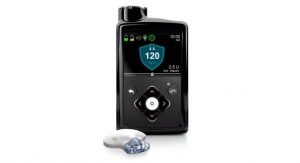
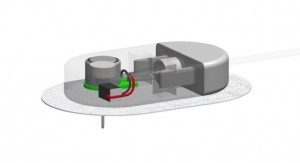
The BD FlowSmart technology consists of a side-ported catheter designed to improve insulin flow and limit the number of flow interruptions that occur when there is a continuous rise in pressure lasting at least 30 minutes, company officials said in a news release. BD collaborated with the Juvenile Diabetes Research Foundation (JDRF) and The Leona M. and Harry B. Helmsley Charitable Trust on the research and development of the new infusion set to enhance the use of insulin pumps and improve the treatment of type 1 diabetes. Approximately two thirds of current insulin pump users have been shown to experience insulin flow interruptions that often go undetected by the pump occlusion alarm system.
“We believe that our new infusion set with BD FlowSmart technology is a ground-breaking innovation in diabetes technology that redefines the role of infusion sets in pump therapy by reducing the incidence of silent occlusions,” said Kenneth Miller, worldwide president of BD Medical-Diabetes Care. “We are especially pleased to achieve this milestone in collaboration with JDRF and the Helmsley Charitable Trust, and that our work together may help people better control their diabetes.”
About two thirds of insulin pump users experience flow interruptions that are undetected by their device’s occlusion alarm system, BD executives noted. A clinical study found that patients who use the company’s infusion set experience significantly fewer flow interruptions, the company adds.
“JDRF is excited to see the potential of BD’s new infusion set for people with type 1 diabetes,” said Derek Rapp, JDRF president/CEO. “Experiencing hyperglycemia for even a short period of time can lead to disastrous consequences such as ketoacidosis, and over long periods of time, concerns of serious complications are a major part of living with type 1 diabetes for many people. Having the assurance of this new technology in the arsenal of tools to help relieve the daily burden of type 1 diabetes is very encouraging. It’s a privilege to work with organizations like the Helmsley Charitable Trust and BD in helping to change the understanding and treatment of this disease.”
The product received Health Canada approval in January and currently is awaiting CE mark approval.
“Now more than ever before, we believe that philanthropy is uniquely positioned to partner with industry to help incentivize and catalyze the development of new therapies and treatments for people with type 1 diabetes,” said David Panzirer, trustee of the Helmsley Charitable Trust. “We are thrilled that, together with JDRF, our support for BD has helped move this infusion technology from the early conceptual stages to an FDA-approved device that can have a real, tangible impact on those living with type 1 diabetes every day.”
In a previously presented head-to-head clinical study with a leading infusion set, subjects using the infusion set with BD FlowSmart technology had significantly fewer flow interruptions and less time spent with interrupted flow.
The infusion set most likely will be launched in 2016.