The ICDs were put into use soon after the company received U.S. Food and Drug Administration (FDA) approval for a new phase of its ongoing ProMRI trial. The new phase is investigating the safety of ICDs in the magnetic resonance imaging (MRI) environment, according to the firm. The first investigational scan involving the DX System occurred at St. Mary Medical Center in Langhorne, Pa.
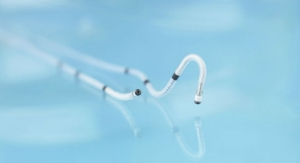
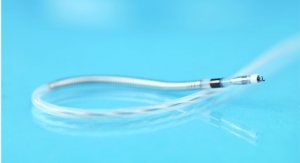
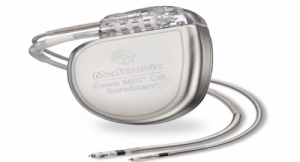
The Biotronik DX ICD system provides physicians with complete diagnostic capabilities of a dual-chamber ICD, including atrial sensing and advanced proven shock reduction algorithms, with only a single lead, the company claims. Biotronik released the DX technology in the United States last year.
“In most cases, it’s difficult for electrophysiologists to accurately predict whether their patients will require diagnostic procedures like MRI in the future,” said Rafael E. Pena, M.D., F.A.C.C., of The Arrhythmia Institute in Newtown, Pa., who performed the ICD implant on the first patient to undergo a full-body MRI scan at St. Mary Medical Center. “Implanting ICDs with the option for MRI allows me to expand their access to this technology when they need it.”
“Frequently, patients with defibrillators need diagnostic imaging with MRI, and until now the safety of MRI imaging in those patients was questioned,” added Thomas Crawford, M.D., assistant professor with the University of Michigan Health System, and a principal investigator of the ProMRI study. “MRI Conditional defibrillators are a major advance in cardiac devices.”
The Biotronik ProMRI study consists of a series of MRI compatibility trials. Phase A resulted in FDA approval of Entovis pacemaker systems for MRI scans with an exclusion zone. Phase B expanded the trial for pacemakers with full-body scans, including cardiac and thoracic spinal scans, and is nearing completion. Now, Phase C incorporates Iforia ICD systems into the list of devices being investigated for full-body MRI scans, including the DX system.
These first U.S. ICD patients receiving full-body scans are among several hundred who already are included in the ongoing study. The FDA approved the ProMRI study expansion to full-body scans for Iforia ICD and DX device systems earlier this year.
Until recently, patients with a pacemaker or ICD were denied MRI scans. The strong forces applied during an MRI scan could negatively affect both implanted device and patient, usually contraindicating MRIs for pacemaker and ICD patients. With its ProMRI technology, Biotronik claims its solution gives all cardiac rhythm patients access to MRI scanning. Investigational full-body scans for DX systems are the next step in expanding U.S. patient access to MRIs.
The Biotronik-sponsored trial (NCT01761162) has been in U.S. subject recruitment since March 2013.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.