Covidien plc has broadened its line of neurovascular products to treat un-ruptured brain aneurysms.
The company recently launched its Pipeline Flex embolization device for the European market at the annual Live Interventional Neuroradiology & Neurosurgery Course, held in Paris, France, June 23-25. The next-generation flow diversion device received CE mark earlier this year.
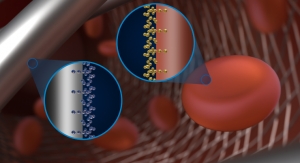
Designed to divert blood flow away from an aneurysm, the Pipeline Flex's design includes a braided cylindrical mesh tube that is implanted across the base or neck of the aneurysm. The device cuts off blood flow to the aneurysm, reconstructing the diseased section of the parent vessel. The device is repositionable and designed for even more accuracy and controlled placement. Among other features, it includes an instant braid release system that, according to the company, makes the device easier to place.
“The Pipeline Flex embolization device is the next advancement in flow diversion, combining our clinically-proven braid design with a new delivery system designed to offer even more accuracy and control when performing these advanced procedures inside the brain,” said Brett Wall, president of Covidien's Neurovascular division. “Covidien is dedicated to seeking customer feedback and advancing product design to meet their needs.”
In Europe, the Pipeline Flex device is intended for the endovascular embolization of cerebral aneurysms. According to the Brain Aneurysm Foundation, there are nearly 500,000 deaths worldwide each year caused by brain aneurysms and half the victims are younger than 50 years of age.
The first-generation Pipeline embolization device has been used to treat patients in Europe since 2009. It has been the only flow diversion device commercially available in the United States since it was approved by the U.S. Food and Drug Administration in April 2011. The Pipeline Flex device is not currently approved for use in the U.S.
Covidien, which is based in Dublin, Ireland, but operates primarily out of U.S. headquarters in Mansfield, Mass., will be merging with industry giant Medtronic Inc. A deal, worth more than $42 billion, was announced a few weeks ago.