Charles Sternberg, Assistant Editor04.21.21
Medtronic plc, a leader in medical technology, has received approval from the United States Food and Drug Administration (FDA) for Pipeline Flex Embolization Device with Shield Technology.
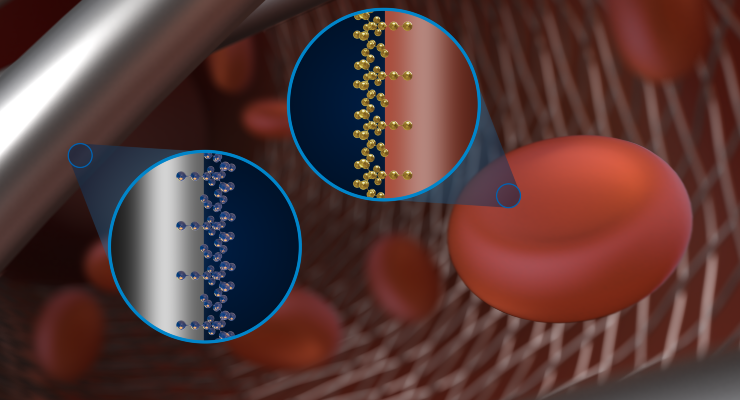
Medtronic developed Shield Technology, a proprietary breakthrough in biomaterial science, to advance flow diversion therapy by introducing a surface-modified implant device which demonstrates reduction in material thrombogenicity, a reduction in the tendency of the surface treatment material to create clots.
NYU Langone Health in New York City performed the first patient procedure in the nation with the new device.
Improving the Pipeline Flex Embolization Device
Celebrating its 10-year anniversary in the U.S. in 2021, Pipeline Flex Embolization Device diverts blood flow away from a brain aneurysm. The first commercially available flow diverter in the market, the device features a braided cylindrical mesh tube that is implanted across the base, or neck, of the aneurysm. The device interrupts blood flow into the aneurysm, reconstructing the diseased section of the parent vessel. Pre-clinical testing shows how Shield Technology improves Pipeline Flex by reducing the thrombogenicity of the device material.
"The Pipeline Flex-Shield that we used today at NYU Langone to treat a giant left internal carotid aneurysm, represents a pivotal milestone in the evolution of flow diversion therapy—establishing a new vanguard for safe and effective management of complex cerebral aneurysms," said Peter Kim Nelson, M.D., chief of Interventional Neuroradiology and professor of Radiology and Neurosurgery at NYU Langone Health. "Our team has long-anticipated the availability of this device for patients in the United States. The surface modification of the implant has demonstrated reduced material thrombogenicity, discernably aiding delivery through tortuous vascular anatomy with improved delivery and resheathing forces compared to earlier generation flow diverters."
Study Results
Published on June 1, 2020, in the Journal of NeuroInterventional Surgery (JNIS), results from the SHIELD Study (Periprocedural to 1-year safety and efficacy outcomes with the Pipeline Embolization Device with Shield technology for intracranial aneurysms: a prospective, post-market, multi-center study) demonstrate that the third-generation Pipeline Flex Embolization Device with Shield Technology continues the strong record of safety and efficacy of flow diversion therapy with the Medtronic family of products. Results show 77.2% complete aneurysm occlusion at 12 months, a 3.2% primary safety endpoint, and 93.1% complete wall apposition post-procedure.
"In addition to addressing material thrombogenicity, Shield Technology improves device performance by reducing the force required for both delivery and resheathing of the device. We believe that Shield Technology opens up a new range of options for physicians to continue to achieve improved patient outcomes," said Dan Volz, president of the Neurovascular business, which is part of the Neuroscience Portfolio at Medtronic. "Ten years ago, Medtronic changed the treatment paradigm in brain aneurysms with the approval of Pipeline Embolization Device, and we continue to challenge ourselves to deliver solutions that improve outcomes and increase value for all healthcare stakeholders."
An estimated 500,000 people throughout the world die each year due to ruptured brain aneurysms, with half the victims younger than 50 years of age. Pipeline Embolization Device has been used to treat patients in the United States since 2011 and is now available with Shield Technology.
"Brain aneurysms are much more common than most people believe and require early detection, accurate diagnosis, and prompt treatment to maximize the chances of survival," said Todd Crawford, founder, Lisa Foundation, dedicated to his wife Lisa who lost her life to complications from a brain aneurysm. "As a valued partner who shared our mission, we commend Medtronic for its continued commitment to pioneering new therapies and approaches to treating brain aneurysms that improve patient outcomes and saves lives."
Medtronic developed Shield Technology, a proprietary breakthrough in biomaterial science, to advance flow diversion therapy by introducing a surface-modified implant device which demonstrates reduction in material thrombogenicity, a reduction in the tendency of the surface treatment material to create clots.
NYU Langone Health in New York City performed the first patient procedure in the nation with the new device.
Improving the Pipeline Flex Embolization Device
Celebrating its 10-year anniversary in the U.S. in 2021, Pipeline Flex Embolization Device diverts blood flow away from a brain aneurysm. The first commercially available flow diverter in the market, the device features a braided cylindrical mesh tube that is implanted across the base, or neck, of the aneurysm. The device interrupts blood flow into the aneurysm, reconstructing the diseased section of the parent vessel. Pre-clinical testing shows how Shield Technology improves Pipeline Flex by reducing the thrombogenicity of the device material.
"The Pipeline Flex-Shield that we used today at NYU Langone to treat a giant left internal carotid aneurysm, represents a pivotal milestone in the evolution of flow diversion therapy—establishing a new vanguard for safe and effective management of complex cerebral aneurysms," said Peter Kim Nelson, M.D., chief of Interventional Neuroradiology and professor of Radiology and Neurosurgery at NYU Langone Health. "Our team has long-anticipated the availability of this device for patients in the United States. The surface modification of the implant has demonstrated reduced material thrombogenicity, discernably aiding delivery through tortuous vascular anatomy with improved delivery and resheathing forces compared to earlier generation flow diverters."
Study Results
Published on June 1, 2020, in the Journal of NeuroInterventional Surgery (JNIS), results from the SHIELD Study (Periprocedural to 1-year safety and efficacy outcomes with the Pipeline Embolization Device with Shield technology for intracranial aneurysms: a prospective, post-market, multi-center study) demonstrate that the third-generation Pipeline Flex Embolization Device with Shield Technology continues the strong record of safety and efficacy of flow diversion therapy with the Medtronic family of products. Results show 77.2% complete aneurysm occlusion at 12 months, a 3.2% primary safety endpoint, and 93.1% complete wall apposition post-procedure.
"In addition to addressing material thrombogenicity, Shield Technology improves device performance by reducing the force required for both delivery and resheathing of the device. We believe that Shield Technology opens up a new range of options for physicians to continue to achieve improved patient outcomes," said Dan Volz, president of the Neurovascular business, which is part of the Neuroscience Portfolio at Medtronic. "Ten years ago, Medtronic changed the treatment paradigm in brain aneurysms with the approval of Pipeline Embolization Device, and we continue to challenge ourselves to deliver solutions that improve outcomes and increase value for all healthcare stakeholders."
An estimated 500,000 people throughout the world die each year due to ruptured brain aneurysms, with half the victims younger than 50 years of age. Pipeline Embolization Device has been used to treat patients in the United States since 2011 and is now available with Shield Technology.
"Brain aneurysms are much more common than most people believe and require early detection, accurate diagnosis, and prompt treatment to maximize the chances of survival," said Todd Crawford, founder, Lisa Foundation, dedicated to his wife Lisa who lost her life to complications from a brain aneurysm. "As a valued partner who shared our mission, we commend Medtronic for its continued commitment to pioneering new therapies and approaches to treating brain aneurysms that improve patient outcomes and saves lives."