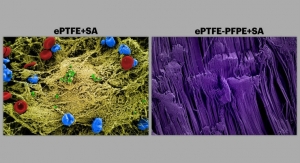
The coating is capable of incorporating multiple pharmaceutical agents and helping preserve the function of various implantable medical devices. Ariste currently is developing a drug-eluting surgical mesh for hernia repair, as well as a drug-eluting vascular graft for the prevention of restenosis-induced graft failure in hemodialysis patients.
The second patent provides additional protection over the method of drug delivery. “We continue to expand and enhance our intellectual property portfolio to ensure the opportunity to commercialize drug-device combination products,” said Ariste Chief Scientific Officer Lisa K. Jennings, Ph.D. “We will be working through the regulatory pathway over the coming year, in pursuit of regulatory clearance in 2015.”
Ariste has completed pre-clinical models demonstrating proof-of-concept using ePTFE grafts coated with antibiotics and has achieved highly significant reductions in bacterial colonization. Ariste claims its technology will help prevent mesh failure due to infection in the hernia repair setting.
In addition, Ariste is pursuing a Series A to fund development of the drug eluting mesh for hernia repair. In 2012, Ariste raised $1.275 million in seed capital.
Ariste Medical Inc. is a privately held biotechnology company developing drug-eluting devices with technology created at the University of Tennessee Health Science Center. Located in the Memphis Bioworks Foundation incubator within the UT-Baptist Research Park in Memphis, Tenn., the company's products are designed to help prevent the failure of surgical devices used in procedures that frequently are compromised by infection, restenosis, or thrombosis