10.28.13
Natick, Mass.-based Boston Scientific Corporation has received the CE mark for its Lotus valve system, a transcatheter aortic valve replacement (TAVR) technology. The system is aimed at providing a treatment alternative for patients with severe aortic stenosis at high risk with surgical valve replacement.
Company officials noted that the Lotus valve system would be immediately available in select centers around Europe. System adoption will accelerate, officials said, as physicians and centers become fully trained.
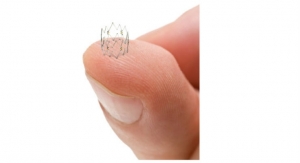
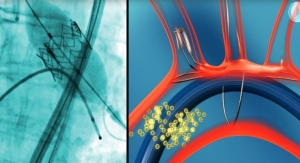
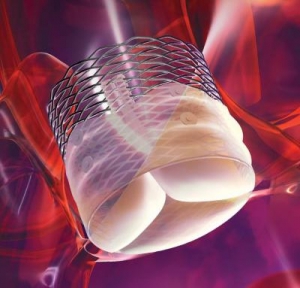
The Lotus valve system is designed to provide physicians increased control during implantation and to help provide a more precise, predictable procedure. It is designed to be assessed in its final position prior to release while at the same time maintaining the ability for the physician to reposition or fully resheath and retrieve the valve. The system also incorporates an adaptive seal technology designed to minimize aortic regurgitation (leaking), a proven predictor of mortality.
“The ability to initially position the Lotus valve precisely and, if needed, to easily reposition or fully retrieve the valve provides the operator with remarkable control,” said Professor Ian Meredith, director of MonashHeart at Monash Medical Centre in Melbourne, Australia, and principal investigator of the REPRISE II trial. “Combined with early and often complete elimination of aortic regurgitation as observed in REPRISE II, the unique features of the Lotus Valve technology represent a significant step forward in the percutaneous treatment of eligible patients with severe symptomatic aortic stenosis.”
“Results from the REPRISE II trial highlight the promise behind the Lotus Valve System, especially related to avoiding moderate or severe paravalvular leaks,” said Nicolas M. Van Mieghem, M.D., of the Erasmus Medical Center in Rotterdam, The Netherlands. “In addition to providing a new treatment option for TAVR, the Lotus Valve has the potential to improve clinical outcomes by minimizing paravalvular leaks.”
“The Lotus Valve System offers patients a new, effective treatment option and provides physicians unmatched positioning and placement capabilities,” said Tom Fleming, vice president and general manager of the structural heart business of Boston Scientific. “It’s the culmination of a decade of research and development and demonstrates our commitment to innovations that make a difference in the lives of patients.”
The Lotus Valve System comes pre-loaded on a transfemoral delivery system which is inserted through a small incision in the leg. Available in a 23 mm and 27 mm size, the system can treat patients with aortic annulus sizes from 20 mm to 27 mm. The Lotus valve system is an investigational device in the United States and Japan and is not available for sale in those countries.
Company officials noted that the Lotus valve system would be immediately available in select centers around Europe. System adoption will accelerate, officials said, as physicians and centers become fully trained.
The Lotus valve system is designed to provide physicians increased control during implantation and to help provide a more precise, predictable procedure. It is designed to be assessed in its final position prior to release while at the same time maintaining the ability for the physician to reposition or fully resheath and retrieve the valve. The system also incorporates an adaptive seal technology designed to minimize aortic regurgitation (leaking), a proven predictor of mortality.
“The ability to initially position the Lotus valve precisely and, if needed, to easily reposition or fully retrieve the valve provides the operator with remarkable control,” said Professor Ian Meredith, director of MonashHeart at Monash Medical Centre in Melbourne, Australia, and principal investigator of the REPRISE II trial. “Combined with early and often complete elimination of aortic regurgitation as observed in REPRISE II, the unique features of the Lotus Valve technology represent a significant step forward in the percutaneous treatment of eligible patients with severe symptomatic aortic stenosis.”
“Results from the REPRISE II trial highlight the promise behind the Lotus Valve System, especially related to avoiding moderate or severe paravalvular leaks,” said Nicolas M. Van Mieghem, M.D., of the Erasmus Medical Center in Rotterdam, The Netherlands. “In addition to providing a new treatment option for TAVR, the Lotus Valve has the potential to improve clinical outcomes by minimizing paravalvular leaks.”
“The Lotus Valve System offers patients a new, effective treatment option and provides physicians unmatched positioning and placement capabilities,” said Tom Fleming, vice president and general manager of the structural heart business of Boston Scientific. “It’s the culmination of a decade of research and development and demonstrates our commitment to innovations that make a difference in the lives of patients.”
The Lotus Valve System comes pre-loaded on a transfemoral delivery system which is inserted through a small incision in the leg. Available in a 23 mm and 27 mm size, the system can treat patients with aortic annulus sizes from 20 mm to 27 mm. The Lotus valve system is an investigational device in the United States and Japan and is not available for sale in those countries.