Business Wire05.10.17
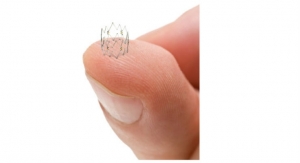
Intact Vascular Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, has hired Howard Rosen as vice president of marketing and business development. In this position, Rosen will lead the company’s efforts to prepare for commercialization of its Tack Endovascular System for the repair of dissections following peripheral artery angioplasty.
“Howard brings deep experience in both upstream and downstream marketing, as well as first-hand knowledge of the peripheral vascular marketplace and our customers,” said Bruce J. Shook, Intact Vascular’s president and CEO. “Howard is a welcome addition to our team as we move through the pre-market approval (PMA) application process and toward market introduction of our products.”
Prior to joining Intact Vascular, Rosen’s career included 21 years at Boston Scientific Corporation, where he began his career as a sales representative and rose through the marketing ranks to hold positions of increasing responsibility in the peripheral vascular interventions and cardiovascular businesses. Rosen most recently served as vice president of the Cell Processing Franchise at Haemonetics Corporation.
“Intact Vascular has created a novel solution to the widespread problem of dissections following angioplasty, and the company has made a significant investment in clinical research. I am excited to build on that strong foundation and bring this important technology to clinicians and their patients,” said Rosen.
Intact Vascular is sponsoring three clinical trials to evaluate its Tack Endovascular System: TOBA II, TOBA II BTK and TOBA III. TOBA II is investigating the combination of the Tack device, with both plain and drug-coated balloon angioplasty in the arteries above the knee. TOBA II BTK is investigating the combination of the Tack device with plain balloon angioplasty in the arteries below the knee. TOBA III is currently underway in Europe and is investigating the combination of the Tack Endovascular System with drug-coated balloon angioplasty.
Intact Vascular is a privately held developer of minimally invasive peripheral vascular products. The Tack Endovascular System is designed to optimize peripheral balloon angioplasty results in the treatment of peripheral arterial disease. The company is based in Wayne, Pa.
“Howard brings deep experience in both upstream and downstream marketing, as well as first-hand knowledge of the peripheral vascular marketplace and our customers,” said Bruce J. Shook, Intact Vascular’s president and CEO. “Howard is a welcome addition to our team as we move through the pre-market approval (PMA) application process and toward market introduction of our products.”
Prior to joining Intact Vascular, Rosen’s career included 21 years at Boston Scientific Corporation, where he began his career as a sales representative and rose through the marketing ranks to hold positions of increasing responsibility in the peripheral vascular interventions and cardiovascular businesses. Rosen most recently served as vice president of the Cell Processing Franchise at Haemonetics Corporation.
“Intact Vascular has created a novel solution to the widespread problem of dissections following angioplasty, and the company has made a significant investment in clinical research. I am excited to build on that strong foundation and bring this important technology to clinicians and their patients,” said Rosen.
Intact Vascular is sponsoring three clinical trials to evaluate its Tack Endovascular System: TOBA II, TOBA II BTK and TOBA III. TOBA II is investigating the combination of the Tack device, with both plain and drug-coated balloon angioplasty in the arteries above the knee. TOBA II BTK is investigating the combination of the Tack device with plain balloon angioplasty in the arteries below the knee. TOBA III is currently underway in Europe and is investigating the combination of the Tack Endovascular System with drug-coated balloon angioplasty.
Intact Vascular is a privately held developer of minimally invasive peripheral vascular products. The Tack Endovascular System is designed to optimize peripheral balloon angioplasty results in the treatment of peripheral arterial disease. The company is based in Wayne, Pa.