Niki Arrowsmith05.30.13
Wayne, N.J.-based Maquet Cardiovascular LLC, a cardiovascular technology company, has acquired Laax Inc., a company based in Livermore, Calif., that makes a mechanical occlusion device named Tigerpaw II. The device is designed to occlude the left atrial appendage (LAA) and conforms to its shape and thickness, helping to minimize the risk of tissue damage and accidental bleeding associated with other common closing methods used during open chest procedures. The product has received 510(k) clearance from the U.S. Food and Drug Administration, CE Mark from the European Union, and will commercially be available in those regions in late spring.
The purchase price was not disclosed.
“We believe Tigerpaw II will be a best-in-class device for reliable ligation of the LAA, potentially mitigating post-operative stroke and its associated costs in patients with atrial fibrillation undergoing cardiac surgery,” said Peter Hinchliffe, president and CEO for the Cardiac Surgery Business Unit of Maquet Cardiovascular. “Atrial fibrillation affects millions of people worldwide, and the Centers for Disease Control and Prevention expect that number to climb significantly in the years to come. We believe Tigerpaw II will play an important role for our customers as they treat more and more patients with atrial fibrillation.”
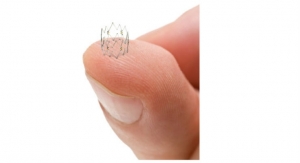
Tigerpaw II consists of a single-use delivery tool and an implantable fastener. The delivery tool has two triggers, one for positioning and placement of the fastener, and the second for release of the fastener, allowing delivery tool removal. The jaws can open to a 15-degree angle. In a clinical trial, the placement of the fastener, timed from hand in to hand off, was 60 seconds or less with the average being 27 seconds. The delivery tool also allows for unlimited placement trials before deployment.
The fastener is made of soft silicone that reportedly conforms to the appendage anatomy and tissue thickness, spreading the pressure equally. According to Laax, Tigerpaw II is the only device currently available that has been proven to be occlusive of the LAA during cardiac surgery due to the pliable silicone. The fastener is designed around the concept of interrupted mattress sutures, which commonly are used in friable, dynamic or compliant tissue environments. They consist of a series of evenly-spaced individual connectors embedded within a soft, compliant elastomeric housing.
“We are continuously evaluating and pursuing technologies that strengthen our offering to customers, and we believe this acquisition allows us to round out our cardiac surgery portfolio and provides us with an entry point into a growing market,” said Raoul Quintero, President and CEO of Maquet Medical Systems USA. “Tigerpaw II is our first product—in what we hope will become a portfolio of products—that helps address the needs of this important patient population.”
Maquet intends to release the device in other regions worldwide after commercializing it in the United States and European Union.
Maquet’s world headquarters is in Rastatt, Germany.
The purchase price was not disclosed.
“We believe Tigerpaw II will be a best-in-class device for reliable ligation of the LAA, potentially mitigating post-operative stroke and its associated costs in patients with atrial fibrillation undergoing cardiac surgery,” said Peter Hinchliffe, president and CEO for the Cardiac Surgery Business Unit of Maquet Cardiovascular. “Atrial fibrillation affects millions of people worldwide, and the Centers for Disease Control and Prevention expect that number to climb significantly in the years to come. We believe Tigerpaw II will play an important role for our customers as they treat more and more patients with atrial fibrillation.”
Tigerpaw II consists of a single-use delivery tool and an implantable fastener. The delivery tool has two triggers, one for positioning and placement of the fastener, and the second for release of the fastener, allowing delivery tool removal. The jaws can open to a 15-degree angle. In a clinical trial, the placement of the fastener, timed from hand in to hand off, was 60 seconds or less with the average being 27 seconds. The delivery tool also allows for unlimited placement trials before deployment.
The fastener is made of soft silicone that reportedly conforms to the appendage anatomy and tissue thickness, spreading the pressure equally. According to Laax, Tigerpaw II is the only device currently available that has been proven to be occlusive of the LAA during cardiac surgery due to the pliable silicone. The fastener is designed around the concept of interrupted mattress sutures, which commonly are used in friable, dynamic or compliant tissue environments. They consist of a series of evenly-spaced individual connectors embedded within a soft, compliant elastomeric housing.
“We are continuously evaluating and pursuing technologies that strengthen our offering to customers, and we believe this acquisition allows us to round out our cardiac surgery portfolio and provides us with an entry point into a growing market,” said Raoul Quintero, President and CEO of Maquet Medical Systems USA. “Tigerpaw II is our first product—in what we hope will become a portfolio of products—that helps address the needs of this important patient population.”
Maquet intends to release the device in other regions worldwide after commercializing it in the United States and European Union.
Maquet’s world headquarters is in Rastatt, Germany.