Maria Shepherd, President and Founder, Medi-Vantage03.15.18
The number of new disease states that can be addressed with implantable neurostimulation devices continues to grow. The most common currently indicated use for these technologies is for the treatment of chronic pain—an enormous market that impacts patient quality of life and productivity. In 2011, it was estimated1 that 100 million U.S. adults suffered from chronic pain, defined as continuous pain every day for the previous three months. Globally, 1.5 billion adults2 suffer from chronic pain (Table 1).
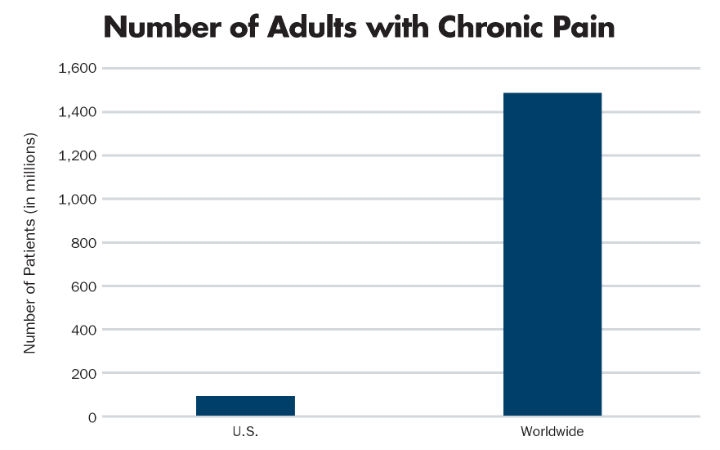
Table 1
Why This Is Important
Even with technology momentum due in part to multiple neurostimulation entrepreneurs, patients suffering from chronic pain continue to be conspicuously underserved. The most obvious patient population is opioid abusers. The CDC estimates that 4.3 million Americans use prescription opioids each month for a non-medical reason, the output of an estimated 249 million opioid pain medication prescriptions written each year.3 Further, 40 people die each day from overdoses involving prescription opioids.3 At the same time, Purdue Pharma, the manufacturer of OxyContin, generated $1.8 billion in 2016.4 The management of chronic pain is a huge market and presents an enormous challenge.
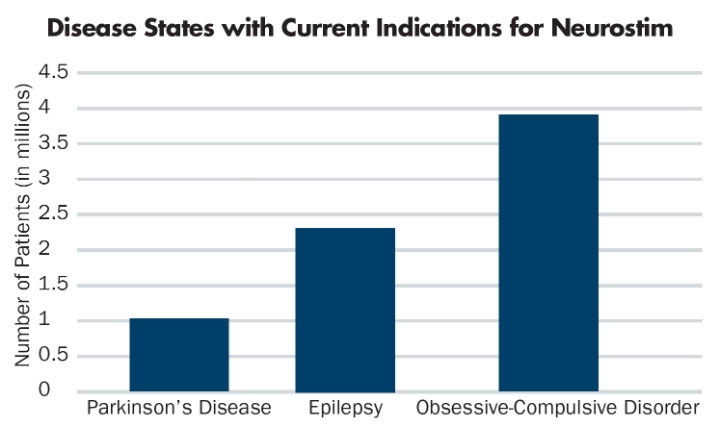
Table 2
In addition to chronic pain, indications for neurostimulation devices include Parkinson’s disease,5 epilepsy,6 and obsessive-compulsive disorder (OCD)7 (Table 2). Future uses cross a broad spectrum of disease states; that’s where this technology and market opportunity gets interesting. Who would have thought that pharmaceutical-focused disease states, such as depression,8 Alzheimer’s,9 migraine headache,10 sleep apnea,11 and tinnitus12 might all respond to a medical device, disrupting the current, drug-based market (Table 3)?
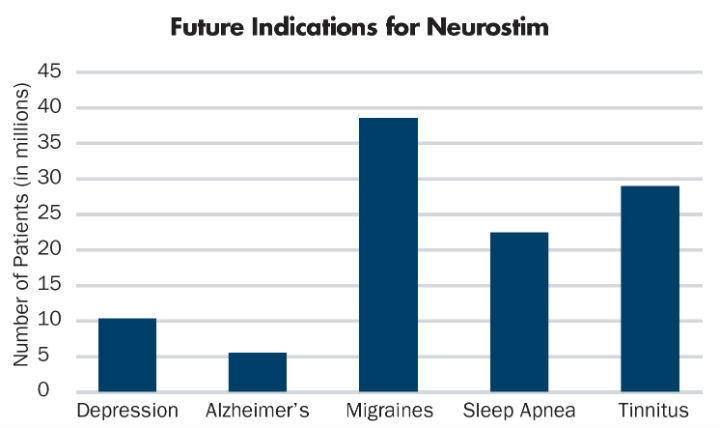
Table 3
It’s a Crowded Market
The top three device types in terms of market size and growth over the next five years are spinal cord stimulators (chronic pain), deep brain stimulation (Parkinson’s disease, epilepsy, and OCD), and sacral nerve stimulation (incontinence). Many startup medical device companies are competing aggressively to gain share in these markets. Despite obstacles such as high device costs, difficult reimbursement, and low awareness of referring physicians, the market for these three types of devices is projected to grow from $4.5 billion in 2015 to $13.7 billion in 202313 (Table 4).
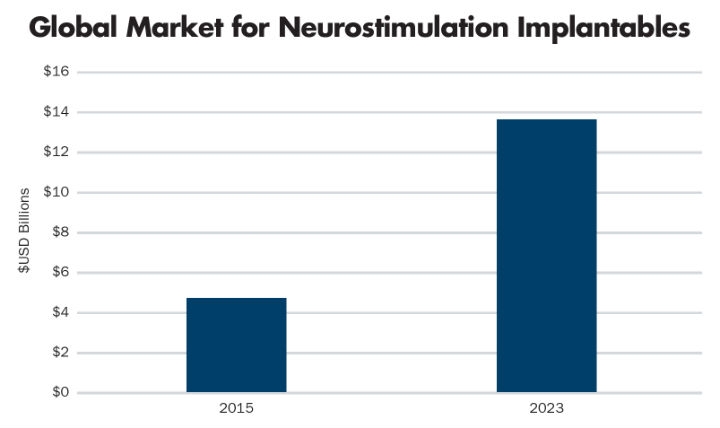
Table 4
The Medi-Vantage Perspective
It’s no wonder Medtronic, St. Jude Medical, and Boston Scientific have brought out the “big guns” to preserve market share in neurostim. They recognize future applications for neurostimulation include some of the largest, most profitable, and challenging disease states.
There are still many unmet needs in implantable neurostimulation devices. Research tells us designs have plenty of room for improvement and the patient experience can be unpleasant during and after the implant procedure. An unpleasant experience may be acceptable to the chronic pain patient who has suffered immensely and seeks relief. As neurostimulation is applied to other disease states, however, the bar as to what is considered acceptable will be raised, especially if a pharmaceutical product is a possible alternative, such as with depression. This market is one where understanding the unmet needs of the end user—the patient—will be especially important.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.

Table 1
Why This Is Important
Even with technology momentum due in part to multiple neurostimulation entrepreneurs, patients suffering from chronic pain continue to be conspicuously underserved. The most obvious patient population is opioid abusers. The CDC estimates that 4.3 million Americans use prescription opioids each month for a non-medical reason, the output of an estimated 249 million opioid pain medication prescriptions written each year.3 Further, 40 people die each day from overdoses involving prescription opioids.3 At the same time, Purdue Pharma, the manufacturer of OxyContin, generated $1.8 billion in 2016.4 The management of chronic pain is a huge market and presents an enormous challenge.

Table 2
In addition to chronic pain, indications for neurostimulation devices include Parkinson’s disease,5 epilepsy,6 and obsessive-compulsive disorder (OCD)7 (Table 2). Future uses cross a broad spectrum of disease states; that’s where this technology and market opportunity gets interesting. Who would have thought that pharmaceutical-focused disease states, such as depression,8 Alzheimer’s,9 migraine headache,10 sleep apnea,11 and tinnitus12 might all respond to a medical device, disrupting the current, drug-based market (Table 3)?

Table 3
It’s a Crowded Market
The top three device types in terms of market size and growth over the next five years are spinal cord stimulators (chronic pain), deep brain stimulation (Parkinson’s disease, epilepsy, and OCD), and sacral nerve stimulation (incontinence). Many startup medical device companies are competing aggressively to gain share in these markets. Despite obstacles such as high device costs, difficult reimbursement, and low awareness of referring physicians, the market for these three types of devices is projected to grow from $4.5 billion in 2015 to $13.7 billion in 202313 (Table 4).

Table 4
The Medi-Vantage Perspective
It’s no wonder Medtronic, St. Jude Medical, and Boston Scientific have brought out the “big guns” to preserve market share in neurostim. They recognize future applications for neurostimulation include some of the largest, most profitable, and challenging disease states.
There are still many unmet needs in implantable neurostimulation devices. Research tells us designs have plenty of room for improvement and the patient experience can be unpleasant during and after the implant procedure. An unpleasant experience may be acceptable to the chronic pain patient who has suffered immensely and seeks relief. As neurostimulation is applied to other disease states, however, the bar as to what is considered acceptable will be raised, especially if a pharmaceutical product is a possible alternative, such as with depression. This market is one where understanding the unmet needs of the end user—the patient—will be especially important.
References
- http://bit.ly/mpo180301
- http://bit.ly/mpo180302
- http://bit.ly/mpo180303 [PDF]
- http://bit.ly/mpo180304
- http://bit.ly/mpo180305
- http://bit.ly/mpo180306
- http://bit.ly/mpo180307
- http://bit.ly/mpo180308
- http://bit.ly/mpo180309
- http://bit.ly/mpo180310
- http://bit.ly/mpo180311
- http://bit.ly/mpo180312
- http://bit.ly/mpo180313
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.