Maria Shepherd , President and Founder, Medi-Vantage11.07.17
AdvaMed exists to advance medical technology around the world as a representative for medical device, diagnostics, and digital health technology companies. Maintaining both a national and global presence in regions and countries including Europe, India, China, Brazil, and Japan, AdvaMed represents medtech organizations from the largest to the smallest medical technology innovators. The AdvaMed annual meeting—The MedTech Conference (held in September every year)—presents a meeting agenda completely dedicated to challenges, trends, regulatory issues, and more within the medical device industry.
Why This Is Important
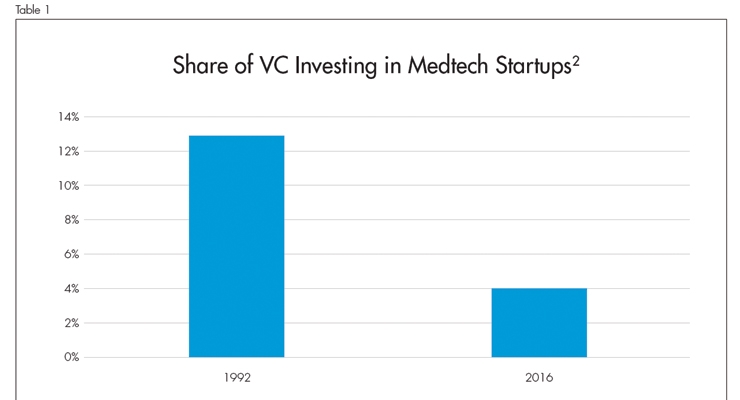
A report from Deloitte and AdvaMed executive leadership1 states the issue clearly: the decrease in medtech startup investments and funding is real and jeopardizes innovation. Medtech startups are the life-blood of innovation in our industry. Without a healthy lineup of acquisition opportunities, large medtech companies will confront challenges in acquiring new technologies to increase and grow. The share of venture capital (VC) investment in medtech companies declined from 13 percent in 1992 to 4 percent in 2016 (Table 1).
Medtech Innovation Is at Risk
Even worse news, the share of VC funding going toward Series A (the first round of VC funds raised by medtech startups) has declined. Investments in medtech Series A funds (as a percentage of total venture investments) sank from 19 percent in 2006 to 10 percent in 2016 (Table 2).

The Medi-Vantage Perspective
It is excruciating to watch the blood, sweat, and tears medtech startup CEOs put into finding funding. Even more painful to watch are the efforts they must make once they cross all the hurdles, have received approval, and launch into their commercialization strategy. The hunt for funding isn’t over until there are sufficient resources to pay for the traditional sales and marketing teams—a significant expense in medtech.
One alternative? We have been helping medtech CEOs commercialize using new, innovative strategies that are less expensive (but still require resources). In some cases, social media can be effective; for others, developing a Center of Excellence that draws patients in can be the right solution. Each medical device startup is different, but strong research and a clear understanding of the target market can provide the keys to success.
How Do We Reverse the Curse?
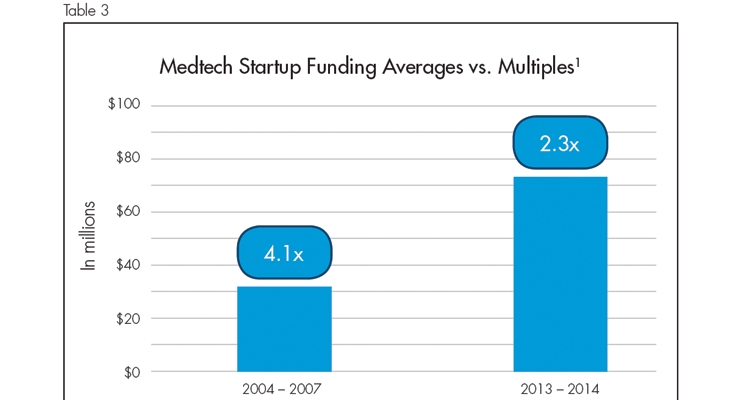
The aforementioned Deloitte report cited an analysis of VC returns conducted by Norwich Ventures. This analysis compared cash multiples to the total investment in venture capital-funded M&A exits from 2004 to 2014. The analysis found that medical device startups raising greater than $40 million gained lower returns than medtech startups where less than $40 million was invested. The initial 25 VC investments in the analysis (between 2004 and 2007) raised an average of $32.2 million with a multiple of 4.1. The 25 deals analyzed between 2013 and 2014 raised an average of $73.5 million but showed a multiple of only 2.3 (Table 3). The increase in funding required to get a company to exit, along with the lower returns (in multiples), demonstrates the challenge facing many medtech startups.
The Deloitte report found that a factor in the lower average return could be due to investments when medtech companies are building commercialization capabilities. Sound familiar? Late-stage investing to build a sales force is not deemed valuable in M&A. Big medical device companies want to buy revenue in a company with proven commercial success. They are less likely to value the sales and marketing infrastructure that built the sales of the medtech startup.
The Critical Role of AdvaMed in Medtech Startups
Without AdvaMed’s support of this report, the root cause of the issues wouldn’t be aggregated, which means fewer solutions could be found. There are multiple possible answers for medtech entrepreneurs. U.S. medtech startup leadership is still undisputed, but the global decline in medtech entrepreneurship continues. This analysis from AdvaMed and Deloitte demonstrates how medical innovation is at risk, and how we all can work together to find a resolution.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Inc., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com
Why This Is Important
A report from Deloitte and AdvaMed executive leadership1 states the issue clearly: the decrease in medtech startup investments and funding is real and jeopardizes innovation. Medtech startups are the life-blood of innovation in our industry. Without a healthy lineup of acquisition opportunities, large medtech companies will confront challenges in acquiring new technologies to increase and grow. The share of venture capital (VC) investment in medtech companies declined from 13 percent in 1992 to 4 percent in 2016 (Table 1).

Medtech Innovation Is at Risk
Even worse news, the share of VC funding going toward Series A (the first round of VC funds raised by medtech startups) has declined. Investments in medtech Series A funds (as a percentage of total venture investments) sank from 19 percent in 2006 to 10 percent in 2016 (Table 2).

The Medi-Vantage Perspective
It is excruciating to watch the blood, sweat, and tears medtech startup CEOs put into finding funding. Even more painful to watch are the efforts they must make once they cross all the hurdles, have received approval, and launch into their commercialization strategy. The hunt for funding isn’t over until there are sufficient resources to pay for the traditional sales and marketing teams—a significant expense in medtech.
One alternative? We have been helping medtech CEOs commercialize using new, innovative strategies that are less expensive (but still require resources). In some cases, social media can be effective; for others, developing a Center of Excellence that draws patients in can be the right solution. Each medical device startup is different, but strong research and a clear understanding of the target market can provide the keys to success.
How Do We Reverse the Curse?
The aforementioned Deloitte report cited an analysis of VC returns conducted by Norwich Ventures. This analysis compared cash multiples to the total investment in venture capital-funded M&A exits from 2004 to 2014. The analysis found that medical device startups raising greater than $40 million gained lower returns than medtech startups where less than $40 million was invested. The initial 25 VC investments in the analysis (between 2004 and 2007) raised an average of $32.2 million with a multiple of 4.1. The 25 deals analyzed between 2013 and 2014 raised an average of $73.5 million but showed a multiple of only 2.3 (Table 3). The increase in funding required to get a company to exit, along with the lower returns (in multiples), demonstrates the challenge facing many medtech startups.

The Deloitte report found that a factor in the lower average return could be due to investments when medtech companies are building commercialization capabilities. Sound familiar? Late-stage investing to build a sales force is not deemed valuable in M&A. Big medical device companies want to buy revenue in a company with proven commercial success. They are less likely to value the sales and marketing infrastructure that built the sales of the medtech startup.
The Critical Role of AdvaMed in Medtech Startups
Without AdvaMed’s support of this report, the root cause of the issues wouldn’t be aggregated, which means fewer solutions could be found. There are multiple possible answers for medtech entrepreneurs. U.S. medtech startup leadership is still undisputed, but the global decline in medtech entrepreneurship continues. This analysis from AdvaMed and Deloitte demonstrates how medical innovation is at risk, and how we all can work together to find a resolution.
References
- http://bit.ly/mpo171201
- Innovation Counselors LLC, Future at Risk Report: Economic Performance, Entrepreneurship, and Venture Capital in the U.S. Medical Technology, AdvaMed Accel, 2016, p.5. http://bit.ly/mpo171202; PDF accessed August 17, 2017.
- Global Data, “Deals Database,” http://bit.ly/mpo171203; website accessed August 21, 2017.
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Inc., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com