10.09.14
New Freedom, Pa.-based Crescent Industries Inc. is now registered by the U.S. Food and Drug Administration (FDA). The company is an employee-owned custom injection molder of plastic components for the medical, pharmaceutical and dental markets.
Manufacturers that are involved in the production and distribution of medical devices intended for commercial distribution in the United States are required to obtain a registration with the FDA. Most firms required to register also are required to list the devices and the activities performed on those devices at that establishment, according to Crescent officials.
The registration is completed once the quality management system is in compliance with the rules established under the FDA’s quality system regulations under FDA 21 CFR (Code of Federal Regulation) part 820 (Medical Devices). Crescent’s quality management system also complies with ISO 9001 and ISO 13485 manufacturing standards.
The company’s leadership views FDA registration as an “opportunity to grow in the medical device manufacturing industry,” and demonstrate to potential customers that the company has the ability to manufacture products that require FDA approval or clearance.
“FDA registration is a logical extension of the component device manufacturing we’ve been doing for over 30 years,” Eric Paules, vice president of operations, told Medical Product Outsourcing. “New and existing customers will be able to work with us as a single source from design concept to distribution of a finished medical device. As our customers’ requirements grow, we want to grow with them, and FDA registration is an important and logical next step in that process.”
Victor Gurany, manager of quality assurance and compliance for Crescent, said FDA registration would put the company in a position of offering—from a contract manufacturing standpoint—not only the manufacturing of components that are distributed to a finished device manufacturer but also to “manufacture finish-good devices or a component/accessory that are packaged or labeled for commercial distribution for health-related purposes to an end-user.”
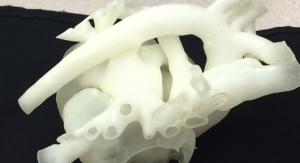
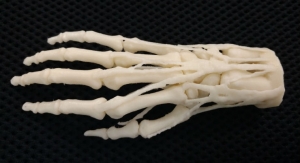
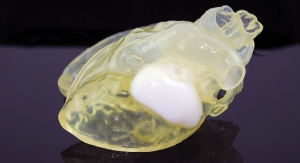
The FDA registration comes soon after Crescent added 3-D printing to its services.
According to company officials, the 3-D printed models have been used to speed up the molding process by using them for design review, quality inspection, product fixtures and end of arm tooling.
The 3-D printing option provides an actual part to set up quality inspection parameters prior to production, eliminating the delay during production for this process. In addition, 3-D printing allows for the manufacture of product fixtures and end-of-arm tooling before production, so there is no waiting for a production quality component to be manufactured before building the fixtures or end of arm tooling, officials noted.
“Crescent’s new Stratasys Eden 500V16 3-D printer gives us the ability to produce ultra-fine details for parts with complex geometries, smooth surface finishes and thin walls,” officials said in a news release. “These components are produced using production grade thermoplastics so painting, gluing, sanding, drilling and bonding with other 3-D printed parts can be completed just like any plastic component.”
The 3-D components can be used as prototypes, samples or even for small production runs.
Founded more than 65 years ago, Crescent offers design and development, mold manufacturing, injection molding and secondary services.
Manufacturers that are involved in the production and distribution of medical devices intended for commercial distribution in the United States are required to obtain a registration with the FDA. Most firms required to register also are required to list the devices and the activities performed on those devices at that establishment, according to Crescent officials.
The registration is completed once the quality management system is in compliance with the rules established under the FDA’s quality system regulations under FDA 21 CFR (Code of Federal Regulation) part 820 (Medical Devices). Crescent’s quality management system also complies with ISO 9001 and ISO 13485 manufacturing standards.
The company’s leadership views FDA registration as an “opportunity to grow in the medical device manufacturing industry,” and demonstrate to potential customers that the company has the ability to manufacture products that require FDA approval or clearance.
“FDA registration is a logical extension of the component device manufacturing we’ve been doing for over 30 years,” Eric Paules, vice president of operations, told Medical Product Outsourcing. “New and existing customers will be able to work with us as a single source from design concept to distribution of a finished medical device. As our customers’ requirements grow, we want to grow with them, and FDA registration is an important and logical next step in that process.”
Victor Gurany, manager of quality assurance and compliance for Crescent, said FDA registration would put the company in a position of offering—from a contract manufacturing standpoint—not only the manufacturing of components that are distributed to a finished device manufacturer but also to “manufacture finish-good devices or a component/accessory that are packaged or labeled for commercial distribution for health-related purposes to an end-user.”
The FDA registration comes soon after Crescent added 3-D printing to its services.
According to company officials, the 3-D printed models have been used to speed up the molding process by using them for design review, quality inspection, product fixtures and end of arm tooling.
The 3-D printing option provides an actual part to set up quality inspection parameters prior to production, eliminating the delay during production for this process. In addition, 3-D printing allows for the manufacture of product fixtures and end-of-arm tooling before production, so there is no waiting for a production quality component to be manufactured before building the fixtures or end of arm tooling, officials noted.
“Crescent’s new Stratasys Eden 500V16 3-D printer gives us the ability to produce ultra-fine details for parts with complex geometries, smooth surface finishes and thin walls,” officials said in a news release. “These components are produced using production grade thermoplastics so painting, gluing, sanding, drilling and bonding with other 3-D printed parts can be completed just like any plastic component.”
The 3-D components can be used as prototypes, samples or even for small production runs.
Founded more than 65 years ago, Crescent offers design and development, mold manufacturing, injection molding and secondary services.