06.10.14
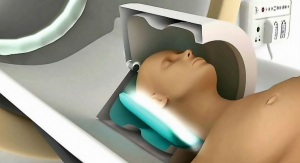
Minneapolis, Minn.-based Medtronic Inc. has received the CE mark for its Advisa and Ensura SR MRI Surescan single-chamber pacemaker devices, and has released the products in Europe.
Both pacemakers are approved for magnetic resonance imaging (MRI) scans positioned on any region of the body.
The devices are not yet approved in the United States.
MRI is the standard of care in soft-tissue imaging, providing information not seen with X-ray, ultrasound, or computed tomography scan, and without exposing patients to ionizing radiation. MRI, therefore, is critical for the early detection, diagnosis and treatment of many diseases, including stroke, cancer, Alzheimer’s disease, muscle, bone and back pain—all of which are prevalent among older adults.
Until the approval of MR-conditional pacemakers, patients with these and other kinds of implanted devices were denied access to MRI procedures because of the potential for harmful interaction between the device and the MRI scanner.
Most other electronic and metal devices, such as brain stimulators or cochlear implants, are dangerous to use in an MRI machine, since the devices can heat up, or move and tear the tissue when placed in MRI machines’ strong magnetic field.
An estimated 50-75 percent of patients with an implantable cardiac device will need an MRI scan over the lifetime of their device, according to figures cited by Medtronic.
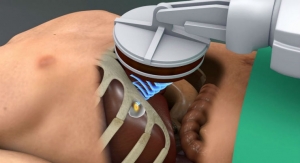
Advisa and Ensura deliver single-chamber pacing engineered with the same Surescan technology used in other Medtronic MR-conditional pacemakers and implantable cardioverter defibrillators (ICDs), while adding features such as improved diagnostic information and storage, as well as a 35 percent improvement in battery longevity (when compared with the company’s Adapta single-chamber pacing system), according to Medtronic.
Pat Mackin, president of Medtronic’s Cardiac Rhythm Disease Management business, said the company has a track record of creating MRI-friendly technology.
“Medtronic has remained committed to developing devices that are approved for MRI since our release of the world’s first MR-conditional pacing system in 2008, and still today we offer the only pacemaker system FDA-approved for MRI scans positioned on any region of the body,” he said.
Both pacemakers are approved for magnetic resonance imaging (MRI) scans positioned on any region of the body.
The devices are not yet approved in the United States.
MRI is the standard of care in soft-tissue imaging, providing information not seen with X-ray, ultrasound, or computed tomography scan, and without exposing patients to ionizing radiation. MRI, therefore, is critical for the early detection, diagnosis and treatment of many diseases, including stroke, cancer, Alzheimer’s disease, muscle, bone and back pain—all of which are prevalent among older adults.
Until the approval of MR-conditional pacemakers, patients with these and other kinds of implanted devices were denied access to MRI procedures because of the potential for harmful interaction between the device and the MRI scanner.
Most other electronic and metal devices, such as brain stimulators or cochlear implants, are dangerous to use in an MRI machine, since the devices can heat up, or move and tear the tissue when placed in MRI machines’ strong magnetic field.
An estimated 50-75 percent of patients with an implantable cardiac device will need an MRI scan over the lifetime of their device, according to figures cited by Medtronic.
Advisa and Ensura deliver single-chamber pacing engineered with the same Surescan technology used in other Medtronic MR-conditional pacemakers and implantable cardioverter defibrillators (ICDs), while adding features such as improved diagnostic information and storage, as well as a 35 percent improvement in battery longevity (when compared with the company’s Adapta single-chamber pacing system), according to Medtronic.
Pat Mackin, president of Medtronic’s Cardiac Rhythm Disease Management business, said the company has a track record of creating MRI-friendly technology.
“Medtronic has remained committed to developing devices that are approved for MRI since our release of the world’s first MR-conditional pacing system in 2008, and still today we offer the only pacemaker system FDA-approved for MRI scans positioned on any region of the body,” he said.