PR Newswire02.02.17
QT Vascular Ltd, a global company engaged in the design, assembly, and distribution of advanced therapeutic solutions for the minimally invasive treatment of vascular disease, announced today that it has signed a definitive agreement with Medtronic Inc. for the worldwide distribution of its Chocolate PTA catheter for a period of five years and automatically renewable for two additional one-year periods. The parties continue to negotiate other aspects of their commercial relationship.
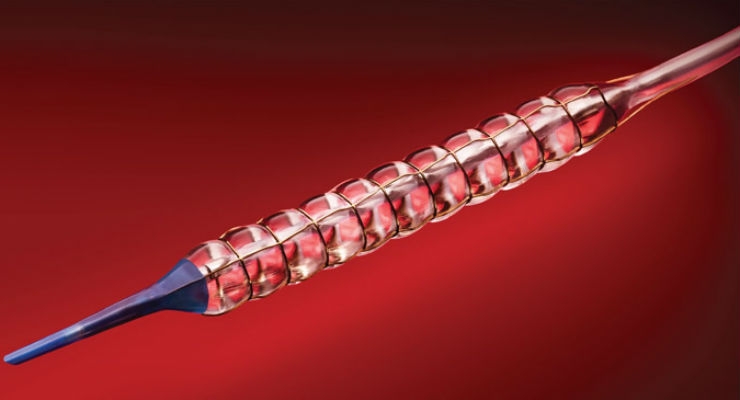
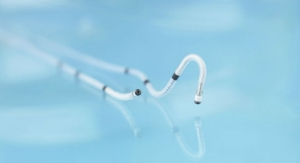
Chocolate represents a breakthrough in PTA design. Its unique nitinol constraining structure is designed to provide atraumatic dilatation in the treatment of blocked arteries. The Chocolate BAR1 post-market registry of 490 patients showed that use of Chocolate PTA resulted in low rates of dissections and bailout stenting. Chocolate PTA may be used as a stand-alone treatment or adjunctive treatment for stenosis in vessels above and below the knee. It is approved for use in the United States, Europe, Australia, Turkey, Singapore and Hong Kong.
"We are honored to partner with Medtronic, the world's leading medical device company. Through our direct and indirect sales channels, the adoption of Chocolate PTA has expanded steadily over the past several years. It is now available in hundreds of hospitals worldwide, and we expect Medtronic's global commercial capabilities will further expand the reach of this device both inside and outside the United States," stated Eitan Konstantino, PhD, CEO of QT Vascular, "Our other products continue to attract interest and we are evaluating additional M&A transactions and strategic options to further enhance value."
Current sales of Chocolate PTA in the United States are conducted through the direct sales force of the Company's wholly-owned subsidiary, TriReme Medical LLC, and a distribution partner. Outside the United States, sales are conducted via country-specific distribution partners. Under the new distribution agreement with Medtronic, transition of the Chocolate PTA business will occur on a country-by-country basis over the coming months starting with the United States.
The Agreement with Medtronic is not expected to have any material effect on the earnings/loss per share and net tangible assets/liabilities per share of the Group for the financial year ending 31 December 2017.
In addition to the Agreement and further to the Company's update announcement dated 26 January 2017 the parties continue to negotiate other aspects of their commercial relationship. The Company plans to focus on its drug coated Chocolate program, which demonstrated positive clinical data to date2 and have full IDE approval from the United States Food and Drug Administration.
None of the Company's Directors, substantial shareholders and/or any of their respective associates has any interest, direct or indirect (other than through their shareholdings in the Company), in the Agreement.
The Company will continue to keep shareholders updated of any material developments as and when appropriate, In the meantime, shareholders and potential investors should exercise caution when trading in the shares of the Company as there is no certainty that the negotiations with Medtronic will materialize into additional definitive transaction(s).
References
1Chocolate BAR interim results presented by Mustapha (AMP, August 2015)
2ENDURE trial interim results presented by Holden (LINC, January 2016) )
Chocolate represents a breakthrough in PTA design. Its unique nitinol constraining structure is designed to provide atraumatic dilatation in the treatment of blocked arteries. The Chocolate BAR1 post-market registry of 490 patients showed that use of Chocolate PTA resulted in low rates of dissections and bailout stenting. Chocolate PTA may be used as a stand-alone treatment or adjunctive treatment for stenosis in vessels above and below the knee. It is approved for use in the United States, Europe, Australia, Turkey, Singapore and Hong Kong.
"We are honored to partner with Medtronic, the world's leading medical device company. Through our direct and indirect sales channels, the adoption of Chocolate PTA has expanded steadily over the past several years. It is now available in hundreds of hospitals worldwide, and we expect Medtronic's global commercial capabilities will further expand the reach of this device both inside and outside the United States," stated Eitan Konstantino, PhD, CEO of QT Vascular, "Our other products continue to attract interest and we are evaluating additional M&A transactions and strategic options to further enhance value."
Current sales of Chocolate PTA in the United States are conducted through the direct sales force of the Company's wholly-owned subsidiary, TriReme Medical LLC, and a distribution partner. Outside the United States, sales are conducted via country-specific distribution partners. Under the new distribution agreement with Medtronic, transition of the Chocolate PTA business will occur on a country-by-country basis over the coming months starting with the United States.
The Agreement with Medtronic is not expected to have any material effect on the earnings/loss per share and net tangible assets/liabilities per share of the Group for the financial year ending 31 December 2017.
In addition to the Agreement and further to the Company's update announcement dated 26 January 2017 the parties continue to negotiate other aspects of their commercial relationship. The Company plans to focus on its drug coated Chocolate program, which demonstrated positive clinical data to date2 and have full IDE approval from the United States Food and Drug Administration.
None of the Company's Directors, substantial shareholders and/or any of their respective associates has any interest, direct or indirect (other than through their shareholdings in the Company), in the Agreement.
The Company will continue to keep shareholders updated of any material developments as and when appropriate, In the meantime, shareholders and potential investors should exercise caution when trading in the shares of the Company as there is no certainty that the negotiations with Medtronic will materialize into additional definitive transaction(s).
References
1Chocolate BAR interim results presented by Mustapha (AMP, August 2015)
2ENDURE trial interim results presented by Holden (LINC, January 2016) )