09.13.13
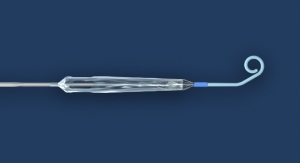
The Ministry of Health, Labor and Welfare in Japan has approved Abbott Laboratories’ Xience Xpedition everolimus-eluting coronary stent system for the treatment of coronary artery disease (CAD), the most common form of heart disease. Xience Xpedition features a new delivery system designed to navigate smoothly through clogged blood vessels in the heart during an angioplasty procedure, particularly in patients with complicated heart vessel anatomy.
Xience Xpedition employs a specialized balloon designed to help physicians open difficult-to-treat blockages and ensures the stent fits securely against the walls of the blood vessel once implanted, according to the company. The catheter that carries the stent has a low-profile design that allows it to move smoothly through the body and heart to the site of the blockage.
The Xience family of stents has been extensively studied in Japanese patients. In addition, several other studies, including investigator-initiated trials, have been conducted since the original launch in Japan of Xience V in 2010.
Xience Xpedition employs a specialized balloon designed to help physicians open difficult-to-treat blockages and ensures the stent fits securely against the walls of the blood vessel once implanted, according to the company. The catheter that carries the stent has a low-profile design that allows it to move smoothly through the body and heart to the site of the blockage.
The Xience family of stents has been extensively studied in Japanese patients. In addition, several other studies, including investigator-initiated trials, have been conducted since the original launch in Japan of Xience V in 2010.