07.23.08
$17 Billion ($172.7B total)
Jeffrey R. Immelt, Chairman and CEO
Joseph M. Hogan, President and CEO, GE Healthcare
Mark L. Vachon, President and CEO, Global Diagnostic Imaging,
GE Healthcare
Omar S. Ishrak, President and CEO, Clinical Systems, GE Healthcare
Peter McCabe, CEO, Surgery
John R. Chiminski, President and CEO, Medical Diagnostics
In the face of FDA warning letters and cuts in imaging reimbursements by Medicare, GE Healthcare posted relatively modest gains in 2007. Though the year was a mixed bag, the healthcare imaging giant still holds on to the No. 2 spot on MPO’s list of Top 30 performing companies. The imaging industry has been fighting reimbursement cuts that were part of the Deficit Reduction Act of 2005, which President Bush signed into law in 2006.
GE Healthcare, based in Chalfont St. Giles, United Kingdom, is a leading maker of diagnostic imaging equipment, such as magnetic resonance imaging (MRI), ultrasound and computed tomography scanners. The company also makes cardiology diagnostic equipment as well as contrast agents used in imaging procedures. In addition, GE Healthcare manufactures clinical equipment such as patient monitors and ventilators; develops life-sciences technology for drug discovery; makes clinical and financial management software for healthcare providers; and provides a range of services from consulting to equipment financing for hospitals.
The company’s revenues rose 3% to $17 billion in 2007. Profit of $3.1 billion in 2007 was 3% lower than in 2006. These numbers are well short of the $20 billion in revenue and $4 billion in profit the company had predicted for 2007. The company said increased sales in the international diagnostic imaging, clinical systems and life-sciences businesses were partially offset by price pressures on US equipment sales and lower sales of surgical imaging equipment resulting from regulatory suspensions of equipment shipments. In January 2007, GE Healthcare executives signed a consent decree with the FDA prohibiting the manufacture and distribution of specified GE OEC Medical Systems X-ray surgical imaging systems at facilities in Salt Lake City, UT and Lawrence, MA until the devices and facilities have been shown to be compliant with the FDA’s current good manufacturing practice requirements.
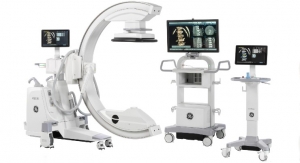
The ban was lifted in early June this year and the FDA said the company had satisfied the agency’s correction requirements. The OEC 9900 Elite C-arm, a fluoroscopy device that uses X-rays to reveal real-time imagery of a patient’s internal structure, is the first product to receive manufacturing and shipping authorization.
“Coming out of this, we’re a better business,” remarked Pete McCabe, president and CEO of GE Healthcare Surgery. “The OEC 9900 C-arm is the most reliable product produced in our business’ 30-year history. Our quality system, alongside customer focus and technical innovations, is a foundation of our business. In the last year, we’ve been able to strengthen it to levels that will ensure we continue to differentiate our products and services as the best in the marketplace. We look forward to again being a leader in this highly competitive field.”
According to the company, every day, seven out of 10 surgeons rely on an OEC C-arm in their operating rooms. In fact, during GE Healthcare Surgery’s stop-ship, many customers held their equipment orders, some waiting more than 18 months, the company claimed. To respond to the demand, GE Healthcare Surgery has added additional shifts and resources to work through its backlog as quickly as possible, company officials said.
In November 2007, the company received another warning from the FDA. The agency sent a letter to the company citing defective devices discovered during FDA field tests that involved two GE Healthcare X-ray devices used in a hospital near New Orleans, LA and assembled at the company’s Pewaukee, WI facility. During the tests, systems were found in which the protective barrier was not in place during the X-ray process to intercept the X-ray beam as required by federal law.
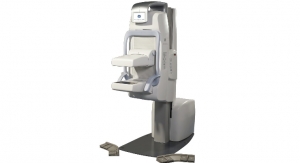
In positive FDA-related news, GE Healthcare received FDA approval in August 2007 for its new mobile mammography for breast cancer screening. The mobile Senographe Essential is built on the company’s Senographe Essential platform, the next generation of GE’s full-field digital mammography systems. In May, the company received FDA clearance for its Carescape Patient Data Module for mobile patient monitoring.
To bolster the company’s inorganic growth and broaden its diagnostic offerings, GE had announced plans in January 2007 to acquire Abbott’s in-vitro and point-of-care diagnostic businesses for $8.13 billion. By July, however, the deal had fallen apart and the companies, whose boards both approved the merger, said they were unable to agree on final terms. In other merger news, GE Healthcare completed the acquisition of Allendale, NJ-based Dynamic Imaging, a developer of Web-based image and information management. The purchase will allow GE to expand its healthcare information technology offerings. Terms of the deal were not disclosed. The acquisition builds on GE Healthcare’s strategy to combine early diagnosis with information technology to enable a new “early health” model of care focused on earlier diagnosis, pre-symptomatic disease detection and disease prevention. The early health theme is one often cited by GE executives.
“Predict, diagnose, treat and monitor is common medical practice today, but now we don’t have to wait so long,” said Joe Hogan, senior vice president and CEO of GE Healthcare, during a meeting with press, industry analysts and physicians in May 2007. “The diagnostic tools are available today, and we can substantially change the overall system.”
GE Chairman and CEO Jeffrey Immelt agreed.
“By doing a better job on diagnostics, you can do a better job of therapy. And that’s where we’re investing,” Immelt said during the same meeting. “High-field imaging, PET [positron emission tomography] scanning and other imaging tools are getting more effective. We’re working on the overall patient experience. We’re also working on taking more devices and imaging tools into the home.”
Immelt also said emerging markets would be another vehicle for GE Healthcare to expand its business. He noted China and India as the biggest emerging markets but also said the Middle East and Africa are changing even more rapidly. He said the company is building plants to manufacture healthcare products in Saudi Arabia to address a projected $20 billion market in the region by 2010.
For the first quarter of 2008, GE Healthcare had revenues of $3.9 billion for the three months ended March 31—almost identical to the same period last year. The firm said that the market remained volatile, with orders for diagnostic imaging down by 13% in the first quarter. However, GE remained optimistic, with international markets posting diagnostic imaging revenues that increased by 8%. Life-sciences sales in Europe were up by 24%. Overall profits for the quarter dropped 17% to $528 million from $637 million in 2007.
“Our focus on globalization has helped sustain the company during the US slowdown,” said Immelt.
Unfortunately, due to declining demand for medical imaging services, the company was forced to lay off hundreds of employees from its Waukesha, WI operations in June this year. GE Healthcare spokesman Brian McKaig said the layoffs would affect fewer than 400 employees, but he did not provide specific totals. McKaig said the medical imaging equipment manufacturer has been hurt by passage of the federal Deficit Reduction Act, which caps Medicare reimbursement levels for medical imaging procedures. McKaig also said in a statement that governments, insurance companies and state legislatures are taking a number of actions aimed at reducing the amount spent on imaging.
In other noteworthy personnel news, Mark Kramer, the former director of the Office of Combination Products at the FDA who had nearly two decades of experience at the agency, joined GE Healthcare in June 2007 as its new vice president of Regulatory Affairs. Kramer is tasked with leading the regulatory function for medical devices across GE Healthcare and will work closely with global regulatory bodies to formulate strategic business plans.
KEY EXECUTIVES:
Jeffrey R. Immelt, Chairman and CEO
Joseph M. Hogan, President and CEO, GE Healthcare
Mark L. Vachon, President and CEO, Global Diagnostic Imaging,
GE Healthcare
Omar S. Ishrak, President and CEO, Clinical Systems, GE Healthcare
Peter McCabe, CEO, Surgery
John R. Chiminski, President and CEO, Medical Diagnostics
NO. OF EMPLOYEES:
46,000 (319,000)GLOBAL HEADQUARTERS:
Fairfield, CTIn the face of FDA warning letters and cuts in imaging reimbursements by Medicare, GE Healthcare posted relatively modest gains in 2007. Though the year was a mixed bag, the healthcare imaging giant still holds on to the No. 2 spot on MPO’s list of Top 30 performing companies. The imaging industry has been fighting reimbursement cuts that were part of the Deficit Reduction Act of 2005, which President Bush signed into law in 2006.
GE Healthcare, based in Chalfont St. Giles, United Kingdom, is a leading maker of diagnostic imaging equipment, such as magnetic resonance imaging (MRI), ultrasound and computed tomography scanners. The company also makes cardiology diagnostic equipment as well as contrast agents used in imaging procedures. In addition, GE Healthcare manufactures clinical equipment such as patient monitors and ventilators; develops life-sciences technology for drug discovery; makes clinical and financial management software for healthcare providers; and provides a range of services from consulting to equipment financing for hospitals.
The company’s revenues rose 3% to $17 billion in 2007. Profit of $3.1 billion in 2007 was 3% lower than in 2006. These numbers are well short of the $20 billion in revenue and $4 billion in profit the company had predicted for 2007. The company said increased sales in the international diagnostic imaging, clinical systems and life-sciences businesses were partially offset by price pressures on US equipment sales and lower sales of surgical imaging equipment resulting from regulatory suspensions of equipment shipments. In January 2007, GE Healthcare executives signed a consent decree with the FDA prohibiting the manufacture and distribution of specified GE OEC Medical Systems X-ray surgical imaging systems at facilities in Salt Lake City, UT and Lawrence, MA until the devices and facilities have been shown to be compliant with the FDA’s current good manufacturing practice requirements.
The ban was lifted in early June this year and the FDA said the company had satisfied the agency’s correction requirements. The OEC 9900 Elite C-arm, a fluoroscopy device that uses X-rays to reveal real-time imagery of a patient’s internal structure, is the first product to receive manufacturing and shipping authorization.
“Coming out of this, we’re a better business,” remarked Pete McCabe, president and CEO of GE Healthcare Surgery. “The OEC 9900 C-arm is the most reliable product produced in our business’ 30-year history. Our quality system, alongside customer focus and technical innovations, is a foundation of our business. In the last year, we’ve been able to strengthen it to levels that will ensure we continue to differentiate our products and services as the best in the marketplace. We look forward to again being a leader in this highly competitive field.”
According to the company, every day, seven out of 10 surgeons rely on an OEC C-arm in their operating rooms. In fact, during GE Healthcare Surgery’s stop-ship, many customers held their equipment orders, some waiting more than 18 months, the company claimed. To respond to the demand, GE Healthcare Surgery has added additional shifts and resources to work through its backlog as quickly as possible, company officials said.
In November 2007, the company received another warning from the FDA. The agency sent a letter to the company citing defective devices discovered during FDA field tests that involved two GE Healthcare X-ray devices used in a hospital near New Orleans, LA and assembled at the company’s Pewaukee, WI facility. During the tests, systems were found in which the protective barrier was not in place during the X-ray process to intercept the X-ray beam as required by federal law.
In positive FDA-related news, GE Healthcare received FDA approval in August 2007 for its new mobile mammography for breast cancer screening. The mobile Senographe Essential is built on the company’s Senographe Essential platform, the next generation of GE’s full-field digital mammography systems. In May, the company received FDA clearance for its Carescape Patient Data Module for mobile patient monitoring.
To bolster the company’s inorganic growth and broaden its diagnostic offerings, GE had announced plans in January 2007 to acquire Abbott’s in-vitro and point-of-care diagnostic businesses for $8.13 billion. By July, however, the deal had fallen apart and the companies, whose boards both approved the merger, said they were unable to agree on final terms. In other merger news, GE Healthcare completed the acquisition of Allendale, NJ-based Dynamic Imaging, a developer of Web-based image and information management. The purchase will allow GE to expand its healthcare information technology offerings. Terms of the deal were not disclosed. The acquisition builds on GE Healthcare’s strategy to combine early diagnosis with information technology to enable a new “early health” model of care focused on earlier diagnosis, pre-symptomatic disease detection and disease prevention. The early health theme is one often cited by GE executives.
“Predict, diagnose, treat and monitor is common medical practice today, but now we don’t have to wait so long,” said Joe Hogan, senior vice president and CEO of GE Healthcare, during a meeting with press, industry analysts and physicians in May 2007. “The diagnostic tools are available today, and we can substantially change the overall system.”
GE Chairman and CEO Jeffrey Immelt agreed.
“By doing a better job on diagnostics, you can do a better job of therapy. And that’s where we’re investing,” Immelt said during the same meeting. “High-field imaging, PET [positron emission tomography] scanning and other imaging tools are getting more effective. We’re working on the overall patient experience. We’re also working on taking more devices and imaging tools into the home.”
Immelt also said emerging markets would be another vehicle for GE Healthcare to expand its business. He noted China and India as the biggest emerging markets but also said the Middle East and Africa are changing even more rapidly. He said the company is building plants to manufacture healthcare products in Saudi Arabia to address a projected $20 billion market in the region by 2010.
For the first quarter of 2008, GE Healthcare had revenues of $3.9 billion for the three months ended March 31—almost identical to the same period last year. The firm said that the market remained volatile, with orders for diagnostic imaging down by 13% in the first quarter. However, GE remained optimistic, with international markets posting diagnostic imaging revenues that increased by 8%. Life-sciences sales in Europe were up by 24%. Overall profits for the quarter dropped 17% to $528 million from $637 million in 2007.
“Our focus on globalization has helped sustain the company during the US slowdown,” said Immelt.
Unfortunately, due to declining demand for medical imaging services, the company was forced to lay off hundreds of employees from its Waukesha, WI operations in June this year. GE Healthcare spokesman Brian McKaig said the layoffs would affect fewer than 400 employees, but he did not provide specific totals. McKaig said the medical imaging equipment manufacturer has been hurt by passage of the federal Deficit Reduction Act, which caps Medicare reimbursement levels for medical imaging procedures. McKaig also said in a statement that governments, insurance companies and state legislatures are taking a number of actions aimed at reducing the amount spent on imaging.
In other noteworthy personnel news, Mark Kramer, the former director of the Office of Combination Products at the FDA who had nearly two decades of experience at the agency, joined GE Healthcare in June 2007 as its new vice president of Regulatory Affairs. Kramer is tasked with leading the regulatory function for medical devices across GE Healthcare and will work closely with global regulatory bodies to formulate strategic business plans.