Maria Shepherd, President and Founder, Medi-Vantage05.03.18
Global revenues for our sister industry—pharmaceuticals—continues to grow (Table 1). By 2022, worldwide pharmaceutical sales are projected to reach an estimated $1.4 trillion at a CAGR of 6.4 percent from 2001 sales of $390 billion.2 That is impressive and steady growth straight through the recession in 2008 and beyond. How do they do it?
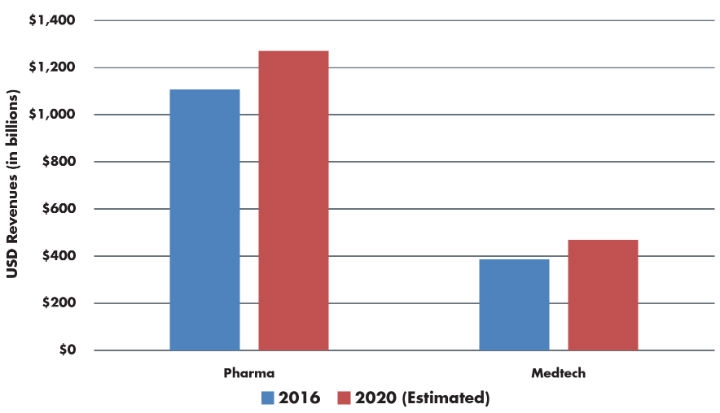
Table 1: A Comparison of Growth—Pharma vs. Medtech1
Why This Is Important
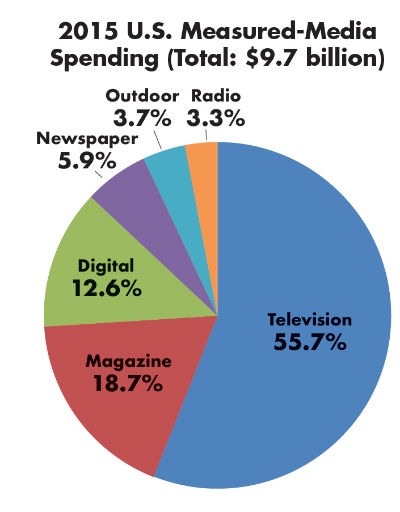
Table 2: Percentage of Healthcare Ad Spending by Medium3
If medtech grew at a CAGR of 6.4 percent, 2020 sales could reach $495 billion—an additional $22 billion in revenue. One reason pharma has grown at (and maintained) a higher CAGR is its significant contribution to the overall healthcare advertising spend in the U.S., which increased to an all-time high of $9.7 billion (up 11 percent).3 New prescription drugs spending, insurers, and increased competition between hospitals have made the healthcare industry one of the 10 top categories for U.S. ad spending. The goal is to create patient demand for these healthcare products. Pharmaceutical-branded advertising was the largest segment of ad spend, totaling $5.7 billion in 2015 (including digital media), up from $3.6 billion in 2012.3
Pharma strategy includes shifting from chemicals to biologics in a bid to stop generic competition. Biologics are genetically created, which, if not replicated by generics, lengthens the branded drugs’ product lifecycles. Although many biologics are targeted toward small numbers of patients, they are reported to be expensive to produce, which means higher costs for consumers and insurers. For example, a course of the Bristol-Myers Squibb immunotherapy drug Opdivo can cost, after discounts, approximately $50,000 a year.3 The list price is reported at $150,000.4
Over 85 percent of the $9.7 billion healthcare media spend is on television, magazines, and digital (Table 2).
Pharma Is a Crowded Market
The top five pharmaceutical companies spend an impressive amount each year to promote their products (Table 3). The branded drugs promoted by advertising make-up a short list. Pharma trends are to carefully place ad spend where it can get the greatest bang for its buck. In 2015, only 16 drugs were promoted in the U.S. with an ad spend of $100 million or more. That number has grown from 2012 when only nine drugs had greater than $100 million budgets.
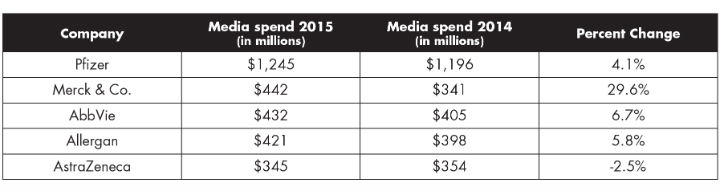
Table 3: Ad Spending of Five Largest Healthcare Marketers3
Legal disclosure requirements are one reason pharma ad spending is high. Pharma magazine ads are usually multi-page, averaging 2.2 pages in 2015. But to supplement their promotional activities with branded drug ads, pharma companies also run unbranded advertising. Unbranded advertising educates on a medical condition and often utilizes celebrity endorsers, who do not mention the specific drug by name. These ads guide patients to the pharma website, to promote the drug that treats the condition. Unbranded pharma ads don’t have the legal disclosures required for branded ads. Unbranded ads were 7 percent of the total pharma ad spend in 2015.3
The Medi-Vantage Perspective
Many of the biologics being advertised are treating small numbers of patients. As shown in the Opdivo ad spend example, the annual cost for a patient can be $50,000 or more. There are many medical devices that approach the $50,000 mark. For example, a transcatheter aortic valve replacement device can cost $32,500.5 The business models for pharma and medtech differ, but spending on the six highest cost medical devices accounts for almost 25 percent of total medical device spend at some hospitals. The devices named in the research were cardiac, orthopedic, and allograft materials. These products address much larger patient populations than those patients indicated for Opdivo. Why not try to create patient demand for medical devices?
If your company is feeling cost-conscious (and who isn’t nowadays?), don’t overlook digital advertising. The number of people in the U.S. using the web as their first stop for health information has reached 42 percent. Twenty-two percent of Americans say they access the internet for healthcare information once a week, at a minimum.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.

Table 1: A Comparison of Growth—Pharma vs. Medtech1
Why This Is Important

Table 2: Percentage of Healthcare Ad Spending by Medium3
Pharma strategy includes shifting from chemicals to biologics in a bid to stop generic competition. Biologics are genetically created, which, if not replicated by generics, lengthens the branded drugs’ product lifecycles. Although many biologics are targeted toward small numbers of patients, they are reported to be expensive to produce, which means higher costs for consumers and insurers. For example, a course of the Bristol-Myers Squibb immunotherapy drug Opdivo can cost, after discounts, approximately $50,000 a year.3 The list price is reported at $150,000.4
Over 85 percent of the $9.7 billion healthcare media spend is on television, magazines, and digital (Table 2).
Pharma Is a Crowded Market
The top five pharmaceutical companies spend an impressive amount each year to promote their products (Table 3). The branded drugs promoted by advertising make-up a short list. Pharma trends are to carefully place ad spend where it can get the greatest bang for its buck. In 2015, only 16 drugs were promoted in the U.S. with an ad spend of $100 million or more. That number has grown from 2012 when only nine drugs had greater than $100 million budgets.

Table 3: Ad Spending of Five Largest Healthcare Marketers3
Legal disclosure requirements are one reason pharma ad spending is high. Pharma magazine ads are usually multi-page, averaging 2.2 pages in 2015. But to supplement their promotional activities with branded drug ads, pharma companies also run unbranded advertising. Unbranded advertising educates on a medical condition and often utilizes celebrity endorsers, who do not mention the specific drug by name. These ads guide patients to the pharma website, to promote the drug that treats the condition. Unbranded pharma ads don’t have the legal disclosures required for branded ads. Unbranded ads were 7 percent of the total pharma ad spend in 2015.3
The Medi-Vantage Perspective
Many of the biologics being advertised are treating small numbers of patients. As shown in the Opdivo ad spend example, the annual cost for a patient can be $50,000 or more. There are many medical devices that approach the $50,000 mark. For example, a transcatheter aortic valve replacement device can cost $32,500.5 The business models for pharma and medtech differ, but spending on the six highest cost medical devices accounts for almost 25 percent of total medical device spend at some hospitals. The devices named in the research were cardiac, orthopedic, and allograft materials. These products address much larger patient populations than those patients indicated for Opdivo. Why not try to create patient demand for medical devices?
If your company is feeling cost-conscious (and who isn’t nowadays?), don’t overlook digital advertising. The number of people in the U.S. using the web as their first stop for health information has reached 42 percent. Twenty-two percent of Americans say they access the internet for healthcare information once a week, at a minimum.
References
- http://bit.ly/mpo180502
- http://bit.ly/mpo180501
- http://bit.ly/mpo180503 [PDF]
- http://bit.ly/mpo180504
- http://bit.ly/mpo180505 [PDF]
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.