Globe Newswire04.02.20
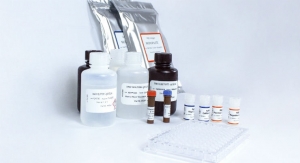
Biomerica Inc. today announced it has signed two separate definitive license agreements with Mount Sinai’s Icahn School of Medicine in New York, N.Y., to license technologies pertaining to a laboratory version serological test for SARS-CoV-2 (“COVID-19”) virus infection which have been developed at Mount Sinai. This test uses the ELISA microplate format that can run on existing open system equipment found in most hospitals and clinical laboratories in the United States.
Biomerica has extensive serological test development, scale-up and manufacturing capabilities and expertise. Based on the data received from Mt. Sinai, the company intends to scale-up commercial manufacturing of the hospital's licensed technology. Biomerica has the equipment and capacity to manufacture more than 1 million tests in the ELISA microplate format per month at its manufacturing facility in Irvine, California. While there are unknowns about the transfer and scale-up process, if successful the company believes it could have commercial sales within weeks. Biomerica will file for expedited clearance with the U.S. Food and Drug Administration (FDA) under the new EUA process for this new test format.
This ELISA microplate format is a serology test that detects antibodies (proteins produced by the immune system in response to infections) in the blood of patients who have been infected with the COVID-19 virus causing the current pandemic. These antibodies show up in detectable quantities in approximately eight days following infection, and remain detectable for as long as three months or more. Persons who have tested positive for the antibodies and are no longer infectious, can possibly be cleared to return to work as they have a lower likelihood of reinfection and/or spreading the virus.
Serology tests are a potentially powerful tool for identifying anyone who has been infected, whether they had symptoms or not. Antibodies to coronaviruses typically remain in humans for up to 90 days or more. Serological testing is important in identifying the total number of people who have been infected with COVID-19 and will allow for further studies that establish whether the immune response protects from future re-infection. Further, this type of testing could be particularly important for the immune surveillance of healthcare workers, first responders, government workers, and others whose infection risks could be heightened by working with COVID-19 infected individuals.
Zackary Irani, chairman and CEO, stated, “If person has already been infected with SARS-CoV-2, they develop antibodies through an immune response that should give them immunity. The entire Biomerica team is working tirelessly to make our low-cost tests available to the hundreds of requests we have received. At the same time, we still remaining committed to our strategy of growing our colorectal disease detection product EZ Detect, and finalizing clinical trials and gaining FDA approval for our HP Detect H. Pylori test and our InFoods IBS diagnostic-guided therapy product.”
Biomerica has extensive serological test development, scale-up and manufacturing capabilities and expertise. Based on the data received from Mt. Sinai, the company intends to scale-up commercial manufacturing of the hospital's licensed technology. Biomerica has the equipment and capacity to manufacture more than 1 million tests in the ELISA microplate format per month at its manufacturing facility in Irvine, California. While there are unknowns about the transfer and scale-up process, if successful the company believes it could have commercial sales within weeks. Biomerica will file for expedited clearance with the U.S. Food and Drug Administration (FDA) under the new EUA process for this new test format.
This ELISA microplate format is a serology test that detects antibodies (proteins produced by the immune system in response to infections) in the blood of patients who have been infected with the COVID-19 virus causing the current pandemic. These antibodies show up in detectable quantities in approximately eight days following infection, and remain detectable for as long as three months or more. Persons who have tested positive for the antibodies and are no longer infectious, can possibly be cleared to return to work as they have a lower likelihood of reinfection and/or spreading the virus.
Serology tests are a potentially powerful tool for identifying anyone who has been infected, whether they had symptoms or not. Antibodies to coronaviruses typically remain in humans for up to 90 days or more. Serological testing is important in identifying the total number of people who have been infected with COVID-19 and will allow for further studies that establish whether the immune response protects from future re-infection. Further, this type of testing could be particularly important for the immune surveillance of healthcare workers, first responders, government workers, and others whose infection risks could be heightened by working with COVID-19 infected individuals.
Zackary Irani, chairman and CEO, stated, “If person has already been infected with SARS-CoV-2, they develop antibodies through an immune response that should give them immunity. The entire Biomerica team is working tirelessly to make our low-cost tests available to the hundreds of requests we have received. At the same time, we still remaining committed to our strategy of growing our colorectal disease detection product EZ Detect, and finalizing clinical trials and gaining FDA approval for our HP Detect H. Pylori test and our InFoods IBS diagnostic-guided therapy product.”