PR Newswire01.24.20
The U.S. Food and Drug Administration (FDA) granted Medtronic plc approval to proceed with an investigational device exemption (IDE) trial to evaluate the safety and effectiveness of the PulseSelect Pulsed Field Ablation (PFA) System, which uses pulsed electric fields to treat atrial fibrillation (AFib). The first procedures in the trial were performed in December 2019.
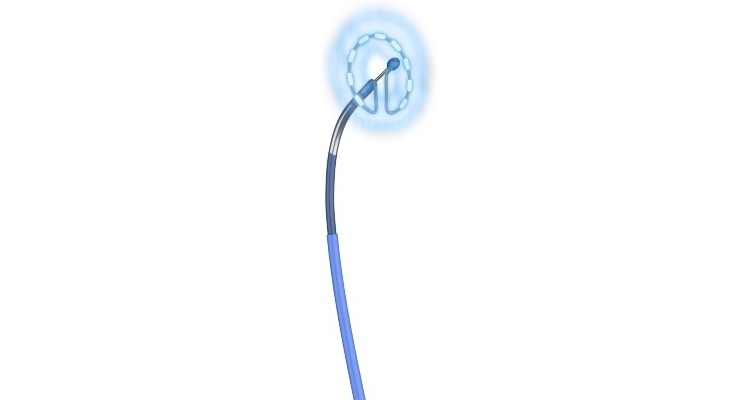
PFA uses pulsed electric fields to ablate or create lesions and scar tissue to interrupt irregular electrical pathways in the heart and the triggers of AFib. However, unlike traditional ablation methods of heating (radio frequency) or cooling (cryo) to ablate the tissue, PFA is non-thermal and selectively targets cardiomyocytes (heart muscle cells) while avoiding other types of tissue.
“This study will evaluate a new energy source that may treat AFib and potentially address the risks that have been associated with other ablation technologies, such as unintended tissue damage,” said Verma. “The rigorous pre-clinical research to get us to these first procedures has been impressive and we are excited to support the development of more clinical evidence.”
In September 2018, the PFA technology was awarded Breakthrough Device designation for the treatment of drug refractory recurrent symptomatic AFib. The system is investigational and not yet approved for sale or distribution.
PULSED AF is a prospective, multi-center, non-randomized, unblinded and worldwide study that will enroll patients who will be treated with the Medtronic PulseSelect PFA System.
“As a global leader in the treatment of cardiac arrythmias, Medtronic is constantly evaluating new and existing therapies to better meet the needs of patients and the physicians who care for them,” said Rebecca Seidel, vice president and general manager in the AFib Solutions division, which is part of the Cardiac and Vascular Group at Medtronic. “The PULSED AF study is another example of our commitment to meaningful innovation and a major step forward in the development of a diverse set of therapy options for AFib patients.”
PFA uses pulsed electric fields to ablate or create lesions and scar tissue to interrupt irregular electrical pathways in the heart and the triggers of AFib. However, unlike traditional ablation methods of heating (radio frequency) or cooling (cryo) to ablate the tissue, PFA is non-thermal and selectively targets cardiomyocytes (heart muscle cells) while avoiding other types of tissue.
“This study will evaluate a new energy source that may treat AFib and potentially address the risks that have been associated with other ablation technologies, such as unintended tissue damage,” said Verma. “The rigorous pre-clinical research to get us to these first procedures has been impressive and we are excited to support the development of more clinical evidence.”
In September 2018, the PFA technology was awarded Breakthrough Device designation for the treatment of drug refractory recurrent symptomatic AFib. The system is investigational and not yet approved for sale or distribution.
PULSED AF is a prospective, multi-center, non-randomized, unblinded and worldwide study that will enroll patients who will be treated with the Medtronic PulseSelect PFA System.
“As a global leader in the treatment of cardiac arrythmias, Medtronic is constantly evaluating new and existing therapies to better meet the needs of patients and the physicians who care for them,” said Rebecca Seidel, vice president and general manager in the AFib Solutions division, which is part of the Cardiac and Vascular Group at Medtronic. “The PULSED AF study is another example of our commitment to meaningful innovation and a major step forward in the development of a diverse set of therapy options for AFib patients.”