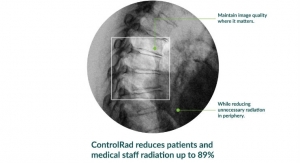
Business Wire11.04.19
Cardiovascular Systems Inc., a medical device company developing and commercializing interventional treatment systems for patients with peripheral and coronary artery disease, has received U.S. Food and Drug Administration (FDA) PMA approval of the new ViperWire Advance Coronary Guide Wire with Flex Tip (ViperWire Advance with Flex Tip).
ViperWire Advance with Flex Tip is the latest advancement to CSI’s coronary Diamondback 360 orbital atherectomy system. Diamondback is the only atherectomy device to treat calcific coronary artery disease by both safely reducing superficial lesions and creating fractures of deep calcium, facilitating stent delivery, expansion and wall apposition for low restenosis rates.
ViperWire Advance with Flex Tip offers a combination of flexible nitinol core with a shapeable floppy tip to help physicians navigate in complex anatomy. In addition, the flexible nitinol body can reduce wire bias during atherectomy. Together, these benefits can enable physicians to access and effectively treat challenging lesions.
“We are treating increasingly complex coronary disease in older patients with more comorbid conditions,” commented Dr. Richard Shlofmitz, chairman, Department of Cardiology at St. Francis Hospital in Roslyn, N.Y. “Many of these patients have calcified coronary lesions which can lead to poor outcomes. Enhancing trackability and performance of atherectomy with ViperWire Advance with Flex Tip will help me more effectively treat calcific disease.”
Dr. Mitchell Krucoff, professor of Medicine at Duke University Medical Center in Durham, N.C., said, “With a shapeable floppy tip and flexible nitinol body, ViperWire Advance with Flex Tip will help physicians efficiently gain lesion access in complex anatomy and reduce wire bias during treatment. This can be a game changer for patient care.”
Scott Ward, CSI’s chairman, president and CEO, said, “We remain committed to expanding our portfolio of differentiated products that help physicians deliver improved outcomes for patients with complex coronary artery disease. The U.S. launch of the new ViperWire Advance with Flex Tip is a key addition to our complex coronary offering that includes the Diamondback 360 Coronary Orbital Atherectomy System with GlideAssist®, the Sapphire balloon portfolio, including the only 1.0 mm coronary angioplasty balloon in the U.S. market, and the Teleport microcatheter.”
Cardiovascular Systems Inc., based in St. Paul, Minn., is a medical device company focused on developing and commercializing solutions for treating vascular and coronary disease. The company’s orbital atherectomy system treats calcified and fibrotic plaque in arterial vessels throughout the leg and heart in a few minutes of treatment time, and address many of the limitations associated with existing surgical, catheter and pharmacological treatment alternatives. The FDA granted 510(k) clearance for the use of the Diamondback Orbital Atherectomy System in peripheral arteries in August 2007. In October 2013, the company received FDA approval for the use of the Diamondback Orbital Atherectomy System in coronary arteries. The Stealth 360 Peripheral Orbital Atherectomy System (OAS) received CE Mark in October 2014. Over 470,000 of CSI’s devices have been sold to institutions worldwide.
ViperWire Advance with Flex Tip is the latest advancement to CSI’s coronary Diamondback 360 orbital atherectomy system. Diamondback is the only atherectomy device to treat calcific coronary artery disease by both safely reducing superficial lesions and creating fractures of deep calcium, facilitating stent delivery, expansion and wall apposition for low restenosis rates.
ViperWire Advance with Flex Tip offers a combination of flexible nitinol core with a shapeable floppy tip to help physicians navigate in complex anatomy. In addition, the flexible nitinol body can reduce wire bias during atherectomy. Together, these benefits can enable physicians to access and effectively treat challenging lesions.
“We are treating increasingly complex coronary disease in older patients with more comorbid conditions,” commented Dr. Richard Shlofmitz, chairman, Department of Cardiology at St. Francis Hospital in Roslyn, N.Y. “Many of these patients have calcified coronary lesions which can lead to poor outcomes. Enhancing trackability and performance of atherectomy with ViperWire Advance with Flex Tip will help me more effectively treat calcific disease.”
Dr. Mitchell Krucoff, professor of Medicine at Duke University Medical Center in Durham, N.C., said, “With a shapeable floppy tip and flexible nitinol body, ViperWire Advance with Flex Tip will help physicians efficiently gain lesion access in complex anatomy and reduce wire bias during treatment. This can be a game changer for patient care.”
Scott Ward, CSI’s chairman, president and CEO, said, “We remain committed to expanding our portfolio of differentiated products that help physicians deliver improved outcomes for patients with complex coronary artery disease. The U.S. launch of the new ViperWire Advance with Flex Tip is a key addition to our complex coronary offering that includes the Diamondback 360 Coronary Orbital Atherectomy System with GlideAssist®, the Sapphire balloon portfolio, including the only 1.0 mm coronary angioplasty balloon in the U.S. market, and the Teleport microcatheter.”
Cardiovascular Systems Inc., based in St. Paul, Minn., is a medical device company focused on developing and commercializing solutions for treating vascular and coronary disease. The company’s orbital atherectomy system treats calcified and fibrotic plaque in arterial vessels throughout the leg and heart in a few minutes of treatment time, and address many of the limitations associated with existing surgical, catheter and pharmacological treatment alternatives. The FDA granted 510(k) clearance for the use of the Diamondback Orbital Atherectomy System in peripheral arteries in August 2007. In October 2013, the company received FDA approval for the use of the Diamondback Orbital Atherectomy System in coronary arteries. The Stealth 360 Peripheral Orbital Atherectomy System (OAS) received CE Mark in October 2014. Over 470,000 of CSI’s devices have been sold to institutions worldwide.