Business Wire09.19.19
Motus GI Holdings Inc., announced that the European Patent Office (EPO) has issued patent No. 3082896 titled, “Colon Cleaning Devices and Methods,” which relates to the company’s flagship product, the Pure-Vu System.
“We continue to strengthen our extensive global IP portfolio covering all aspects of the Pure-Vu System. This patent is particularly important as we look to the future expansion into the European market where approximately 5.6 million colonoscopy procedures were performed in 2018,1” commented Tim Moran, CEO of Motus GI. “Additionally, establishing a robust IP portfolio is directly relevant to our plans to establish strategic partnerships to support global commercialization outside the U.S.”
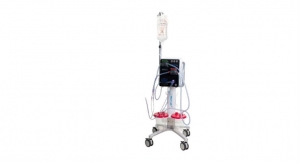
The Pure-Vu System is a 510(k) U.S. Food and Drug Administration cleared medical device indicated to help facilitate the cleaning of a poorly prepared colon during the colonoscopy procedure. The device integrates with standard and slim colonoscopes to enable safe and rapid cleansing during the procedure while preserving established procedural workflow and techniques. The first generation of the Pure-Vu System has received CE mark approval in Europe and Motus GI intends to seek CE Mark approval for the Pure-Vu GEN2 System.
Motus GI Holdings Inc. is a medical technology company, with subsidiaries in the U.S. and Israel, dedicated to improving clinical outcomes and enhancing the cost-efficiency of colonoscopy. The Company’s flagship product is the Pure-Vu System, a U.S. Food and Drug Administration-cleared medical device indicated to help facilitate the cleaning of a poorly prepared colon during the colonoscopy procedure. The device integrates with standard and slim colonoscopes to enable safe and rapid cleansing during the procedure while preserving established procedural workflow and techniques. The first generation of the Pure-Vu System has received CE mark approval in Europe and Motus GI intends to seek CE Mark approval for the Pure-Vu GEN2 System. The Pure-Vu System is currently being introduced on a pilot basis in the U.S. market, and the company is planning to initiate a commercial launch focused on the U.S. hospital market in 2019. Challenges with bowel preparation for inpatient colonoscopy represent a significant area of unmet need that directly affects clinical outcomes and increases the cost of care in a market segment that comprises approximately 1.5 million annual procedures in the U.S. and approximately 4 million annual procedures worldwide. Motus GI believes the Pure-Vu System may improve outcomes and lower costs for hospitals by reducing the time to successful colonoscopy, minimizing delayed and incomplete procedures, and improving the quality of an exam. In clinical studies to date, the Pure-Vu System significantly increased the number of patients with an adequate cleansing level, according to the Boston Bowel Preparation Scale Score, a validated assessment instrument.
Reference
1 IQVIA Holdings, Inc., [2019]
“We continue to strengthen our extensive global IP portfolio covering all aspects of the Pure-Vu System. This patent is particularly important as we look to the future expansion into the European market where approximately 5.6 million colonoscopy procedures were performed in 2018,1” commented Tim Moran, CEO of Motus GI. “Additionally, establishing a robust IP portfolio is directly relevant to our plans to establish strategic partnerships to support global commercialization outside the U.S.”
The Pure-Vu System is a 510(k) U.S. Food and Drug Administration cleared medical device indicated to help facilitate the cleaning of a poorly prepared colon during the colonoscopy procedure. The device integrates with standard and slim colonoscopes to enable safe and rapid cleansing during the procedure while preserving established procedural workflow and techniques. The first generation of the Pure-Vu System has received CE mark approval in Europe and Motus GI intends to seek CE Mark approval for the Pure-Vu GEN2 System.
Motus GI Holdings Inc. is a medical technology company, with subsidiaries in the U.S. and Israel, dedicated to improving clinical outcomes and enhancing the cost-efficiency of colonoscopy. The Company’s flagship product is the Pure-Vu System, a U.S. Food and Drug Administration-cleared medical device indicated to help facilitate the cleaning of a poorly prepared colon during the colonoscopy procedure. The device integrates with standard and slim colonoscopes to enable safe and rapid cleansing during the procedure while preserving established procedural workflow and techniques. The first generation of the Pure-Vu System has received CE mark approval in Europe and Motus GI intends to seek CE Mark approval for the Pure-Vu GEN2 System. The Pure-Vu System is currently being introduced on a pilot basis in the U.S. market, and the company is planning to initiate a commercial launch focused on the U.S. hospital market in 2019. Challenges with bowel preparation for inpatient colonoscopy represent a significant area of unmet need that directly affects clinical outcomes and increases the cost of care in a market segment that comprises approximately 1.5 million annual procedures in the U.S. and approximately 4 million annual procedures worldwide. Motus GI believes the Pure-Vu System may improve outcomes and lower costs for hospitals by reducing the time to successful colonoscopy, minimizing delayed and incomplete procedures, and improving the quality of an exam. In clinical studies to date, the Pure-Vu System significantly increased the number of patients with an adequate cleansing level, according to the Boston Bowel Preparation Scale Score, a validated assessment instrument.
Reference
1 IQVIA Holdings, Inc., [2019]