Johnson & Johnson Medical Devices Companies05.10.19
Johnson & Johnson Medical Devices Companies* announced that Biosense Webster Inc.’s QDOT MICRO, a catheter that facilitates high power-short duration radiofrequency (RF) ablation, demonstrated safety and efficacy in achieving pulmonary vein isolation in patients with symptomatic drug-refractory paroxysmal atrial fibrillation (AF) in QDOT-FAST,** the first in-human multicenter study of the device. Procedure and fluoroscopy times were also reported to be shorter than ablation with conventional catheters. Reducing use of fluoroscopy alleviates the burden of lead for physicians and staff, and reduces exposure to radiation for patients.
The findings were presented at Heart Rhythm 2019, the Heart Rhythm Society's 40th Annual Heart Rhythm Scientific Sessions, by Vivek Y. Reddy, M.D., director of Cardiac Arrhythmia services for The Mount Sinai Hospital and the Mount Sinai Health System and the Helmsley Trust Professor of Medicine in Cardiac Electrophysiology at Icahn School of Medicine at Mount Sinai in New York, N.Y. The study was funded by Biosense Webster.
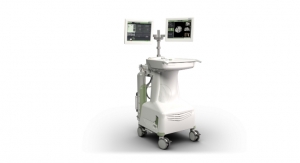
QDOT MICRO, which is only available for investigational use in the United States and Europe, is the first irrigated contact force catheter to deliver up to 90 watts of RF power in up to four seconds in a temperature-controlled ablation mode. The current practice of RF ablation with irrigated catheters involves the delivery of moderate power (20 to 40 watts) for a relatively long duration (20 to 40 seconds).1
In the three-month multicenter prospective study, researchers reported pulmonary vein isolation (PVI) was successful in all 52 paroxysmal AF patients treated with a total average procedure time of 105.2 minutes. PVI was achieved in 44.3 minutes and most fluoroscopy times were between 2.5 and 5.7 minutes. Primary adverse events occurred in 3.8 percent of patients (1 pseudoaneurysm, 1 asymptomatic thromboembolism) and one non-serious adverse event (esophageal ulcer) was device/procedure related.
“The results are very promising and may lead to better patient outcomes and improved procedural efficiencies, including short overall procedure and fluoroscopy times,” said Dr. Reddy,*** study co-author. “Additional prospective studies are needed to assess for the durability of the lesion set and long-term freedom from recurrent atrial arrhythmias.”
“We continue to study QDOT MICRO and generate the evidence necessary to offer a next generation device that we expect to be groundbreaking for catheter ablation procedures,” said Uri Yaron, worldwide president of Biosense Webster. “This study is one of many that we hope elevates the standard of care for atrial fibrillation patients throughout the world.”
In addition to the QDOT-FAST Study, Biosense Webster is also currently enrolling and treating patients in a U.S. Investigational Device Exemption (IDE) study of QDOT MICRO.
An estimated 33 million people worldwide have been diagnosed with AF and its prevalence is projected to increase significantly as the population ages.2 Approximately 70 percent of patients with AF are between the ages of 65 and 85.3
As the world’s most comprehensive medical devices business, Johnson & Johnson Medical Devices Companies are building on a century of experience, leveraging science and technology, to shape the future of healthcare. With unparalleled breadth, depth and reach in surgery, orthopaedics, vision and interventional solutions the organization is working to profoundly change the way care is delivered.
Biosense Webster Inc. is a global leader in the science of diagnosing and treating heart rhythm disorders. The company partners with clinicians to develop innovative technologies that improve the quality of care for arrhythmia patients worldwide. Biosense Webster Inc. is part of the Johnson & Johnson Family of Companies.
*Comprising the surgery, orthopedics, vision and interventional solutions businesses within Johnson & Johnson’s Medical Devices segment
**Pulmonary Vein Isolation with Very High Power–Short Duration Temperature-Controlled Lesions: The First-in-Human QDOT-FAST Multicenter Trial
***Dr. Reddy is a consultant to Biosense Webster Inc.
The device is approved for investigational use only. It is not approved or available for sale.
References
1 http://electrophysiology.onlinejacc.org/content/early/2018/02/02/j.jacep.2017.11.018
2 European Heart Journal, Volume 37, Issue 38, 7 October 2016, Pages 2893–2962, https://doi.org/10.1093/eurheartj/ehw210
3 Amin A, Houmsse A, Ishola A, Tyler J, Houmsse M. The current approach of atrial fibrillation management. Avicenna J Med. 2016 Jan-Mar; 6(1): 8–16.
The findings were presented at Heart Rhythm 2019, the Heart Rhythm Society's 40th Annual Heart Rhythm Scientific Sessions, by Vivek Y. Reddy, M.D., director of Cardiac Arrhythmia services for The Mount Sinai Hospital and the Mount Sinai Health System and the Helmsley Trust Professor of Medicine in Cardiac Electrophysiology at Icahn School of Medicine at Mount Sinai in New York, N.Y. The study was funded by Biosense Webster.
QDOT MICRO, which is only available for investigational use in the United States and Europe, is the first irrigated contact force catheter to deliver up to 90 watts of RF power in up to four seconds in a temperature-controlled ablation mode. The current practice of RF ablation with irrigated catheters involves the delivery of moderate power (20 to 40 watts) for a relatively long duration (20 to 40 seconds).1
In the three-month multicenter prospective study, researchers reported pulmonary vein isolation (PVI) was successful in all 52 paroxysmal AF patients treated with a total average procedure time of 105.2 minutes. PVI was achieved in 44.3 minutes and most fluoroscopy times were between 2.5 and 5.7 minutes. Primary adverse events occurred in 3.8 percent of patients (1 pseudoaneurysm, 1 asymptomatic thromboembolism) and one non-serious adverse event (esophageal ulcer) was device/procedure related.
“The results are very promising and may lead to better patient outcomes and improved procedural efficiencies, including short overall procedure and fluoroscopy times,” said Dr. Reddy,*** study co-author. “Additional prospective studies are needed to assess for the durability of the lesion set and long-term freedom from recurrent atrial arrhythmias.”
“We continue to study QDOT MICRO and generate the evidence necessary to offer a next generation device that we expect to be groundbreaking for catheter ablation procedures,” said Uri Yaron, worldwide president of Biosense Webster. “This study is one of many that we hope elevates the standard of care for atrial fibrillation patients throughout the world.”
In addition to the QDOT-FAST Study, Biosense Webster is also currently enrolling and treating patients in a U.S. Investigational Device Exemption (IDE) study of QDOT MICRO.
An estimated 33 million people worldwide have been diagnosed with AF and its prevalence is projected to increase significantly as the population ages.2 Approximately 70 percent of patients with AF are between the ages of 65 and 85.3
As the world’s most comprehensive medical devices business, Johnson & Johnson Medical Devices Companies are building on a century of experience, leveraging science and technology, to shape the future of healthcare. With unparalleled breadth, depth and reach in surgery, orthopaedics, vision and interventional solutions the organization is working to profoundly change the way care is delivered.
Biosense Webster Inc. is a global leader in the science of diagnosing and treating heart rhythm disorders. The company partners with clinicians to develop innovative technologies that improve the quality of care for arrhythmia patients worldwide. Biosense Webster Inc. is part of the Johnson & Johnson Family of Companies.
*Comprising the surgery, orthopedics, vision and interventional solutions businesses within Johnson & Johnson’s Medical Devices segment
**Pulmonary Vein Isolation with Very High Power–Short Duration Temperature-Controlled Lesions: The First-in-Human QDOT-FAST Multicenter Trial
***Dr. Reddy is a consultant to Biosense Webster Inc.
The device is approved for investigational use only. It is not approved or available for sale.
References
1 http://electrophysiology.onlinejacc.org/content/early/2018/02/02/j.jacep.2017.11.018
2 European Heart Journal, Volume 37, Issue 38, 7 October 2016, Pages 2893–2962, https://doi.org/10.1093/eurheartj/ehw210
3 Amin A, Houmsse A, Ishola A, Tyler J, Houmsse M. The current approach of atrial fibrillation management. Avicenna J Med. 2016 Jan-Mar; 6(1): 8–16.